In which of the following phases are particles of a substance generally closest together?
-
A
Gas
-
B
Liquid
-
C
Plasma
-
D
Solid
Particles of a substance are generally closest together in the solid phase.
The spatial arrangement and average distance between particles (atoms, molecules, or ions) are primary distinguishing features of the states of matter. This distance is governed by the balance between the particles' kinetic energy (which tends to separate them) and the intermolecular forces (which tend to pull them together).
A) Gas
In the gaseous state, particles possess very high kinetic energy. Intermolecular forces are negligible in comparison, allowing particles to move independently and randomly at high speeds. They are widely separated, with large empty spaces between them. Gases are highly compressible because the particles are already so far apart that they can be pushed closer together easily.
B) Liquid
In the liquid state, particles have moderate kinetic energy. They are close enough for intermolecular forces to be significant, giving liquids a definite volume. However, particles have enough energy to slide past one another, allowing flow. The average intermolecular distance is greater than in solids but much smaller than in gases.
C) Plasma
Plasma is often considered the fourth state of matter, occurring at extremely high temperatures where atoms are ionized, creating a soup of free electrons and positive ions. While the charged particles in a dense plasma (like in a star) can be packed together, in most laboratory or observable plasmas (like flames or neon signs), the particles are highly energetic and very far apart, similar to gases. The defining characteristic of plasma is ionization and collective behavior, not necessarily extreme proximity.
D) Solid
In the solid state, particles have the lowest kinetic energy, typically limited to vibrational motion around fixed points in a lattice. Strong intermolecular forces hold the particles in a rigid, highly ordered, and closely packed arrangement. This results in the smallest possible average distance between neighboring particles for a given substance, making solids essentially incompressible and giving them a definite shape and volume.
Conclusion:
The progression from solid to liquid to gas involves increasing particle kinetic energy and decreasing influence of intermolecular forces, leading to greater particle separation. Therefore, particles are most densely packed and closest together in the solid phase.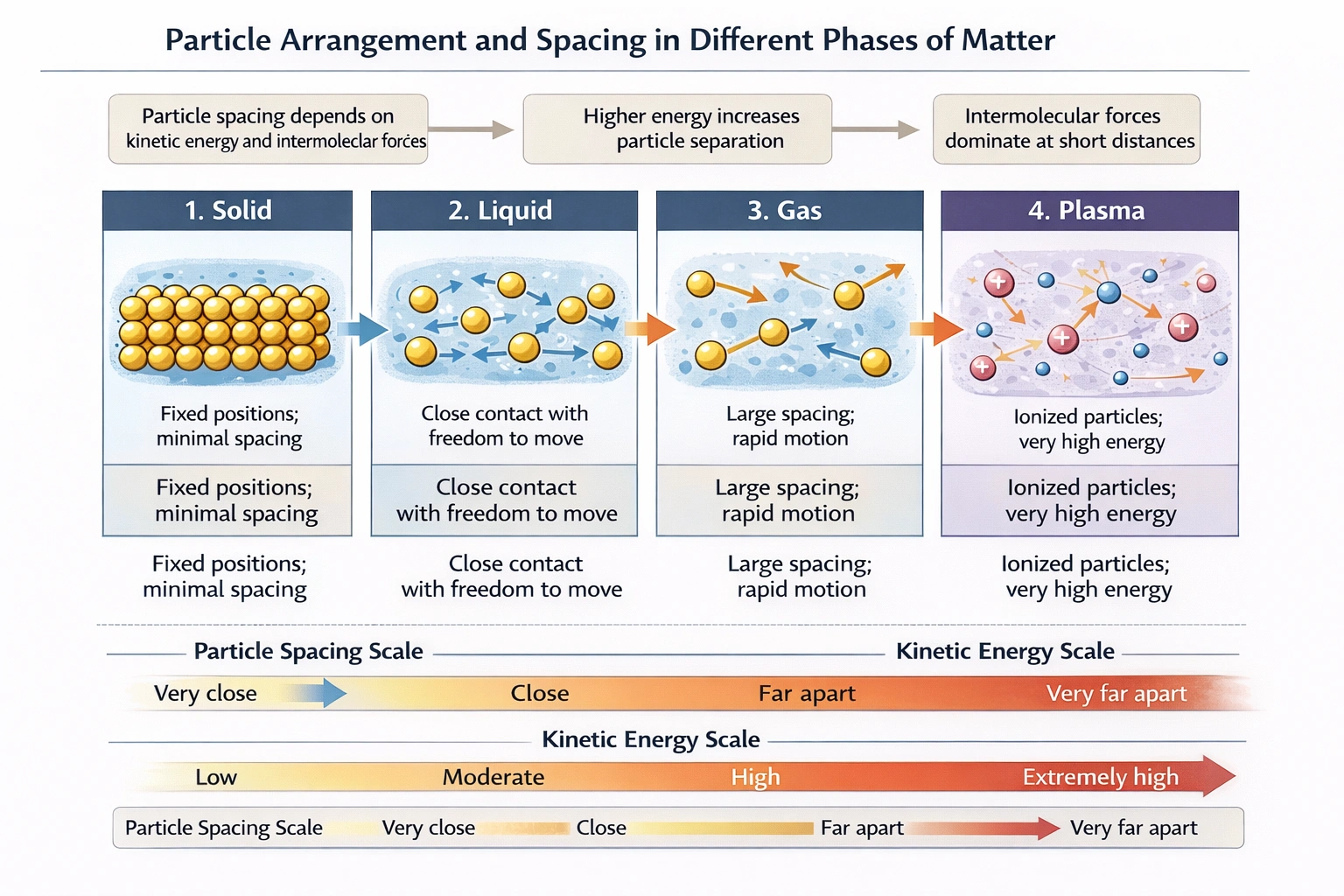
Topic Flashcards
Click to FlipWhich state of matter has particles that are locked in a fixed, closely-packed arrangement with only vibrational motion?
Solid.
Rank the three common states of matter (solid, liquid, gas) in order of DECREASING average distance between particles.
Gas (greatest distance), Liquid, Solid (smallest distance).
What is the primary reason particles in a solid are closer together than in a liquid or gas?
Lower kinetic energy allows stronger intermolecular forces to dominate, holding particles in a tight, ordered structure.
The high compressibility of a gas is direct evidence of what fact about the space between its particles?
There is a great deal of empty space between the widely separated gas particles.
In which state of matter would you expect the intermolecular forces of attraction to have the greatest relative influence on particle behavior?
Solid