Net water movement through a membrane in response to the concentration of a solute is called
-
A
bonding
-
B
diffusion
-
C
osmosis
-
D
polarity
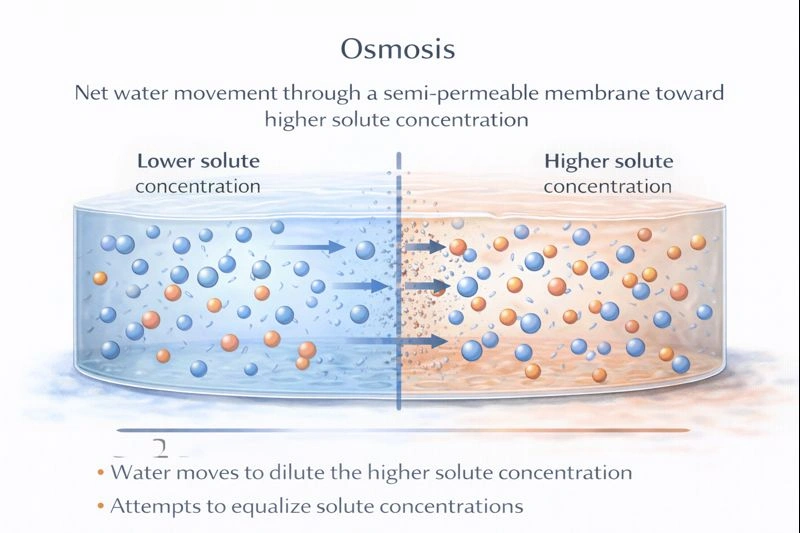
The net movement of water through a membrane in response to a solute concentration gradient is called osmosis.
Osmosis is a special case of diffusion that involves the movement of solvent molecules (typically water) across a selectively permeable membrane. The driving force is the difference in the concentration of solute particles on either side of the membrane. Water moves from the region of lower solute concentration (and higher water concentration) to the region of higher solute concentration (and lower water concentration) in an attempt to equalize the solute concentrations.
A) bonding
Bonding refers to the attractive forces that hold atoms together within molecules (e.g., covalent, ionic bonds) or between molecules (intermolecular forces). It is a static force of attraction, not a description of the net bulk movement of water molecules across a membrane.
B) diffusion
Diffusion is the broader process of net movement of particles (atoms, ions, molecules) from an area of higher concentration to an area of lower concentration. While osmosis is a specific type of diffusion, the term "diffusion" by itself generally refers to the movement of the solute particles themselves through a medium. The question specifically defines the movement of the solvent (water) in response to a solute gradient, which is the precise definition of osmosis.
C) osmosis
Osmosis is defined as the spontaneous net movement of solvent molecules through a selectively permeable membrane into a region of higher solute concentration. This matches the description exactly. It is a critical process in biological systems for maintaining water balance across cell membranes.
D) polarity
Polarity is a molecular property resulting from an uneven distribution of electron density, creating partial positive and negative charges within a molecule. Water's polarity is responsible for its ability to form hydrogen bonds and dissolve many solutes, which underpins the mechanism of osmosis. However, polarity is the cause of the behavior, not the name of the transport process itself.
Conclusion:
Biological and chemical processes require precise terminology. The specific term for the net movement of water across a semipermeable membrane driven by differences in solute concentration is osmosis. It is a passive transport process distinct from general solute diffusion, though related to it.
Topic Flashcards
Click to FlipWhat is the specific term for the net movement of WATER across a semipermeable membrane from a dilute solution to a concentrated one?
Osmosis.
During osmosis, water moves toward the side of the membrane that has a higher concentration of _____.
Solute (dissolved particles).
How is osmosis different from simple diffusion?
Osmosis is the diffusion of the solvent (water) across a membrane in response to a solute gradient, while diffusion typically refers to the movement of the solute particles themselves.
In biological systems, what is the primary function of osmosis related to cells?
To maintain water balance (homeostasis) and turgor pressure.
If a cell is placed in pure water, which way will water move by osmosis, and what might happen to the cell?
Water will move into the cell, potentially causing it to swell or lyse (burst) if it lacks a rigid cell wall.