Which product completes the following acid-base reaction? H₂SO₄ + 2KOH → ?
-
A
H₃O⁺
-
B
HSO₃⁻
-
C
K₂SO₄
-
D
KHSO₄
The products that complete the reaction are K₂SO₄ and water, so K₂SO₄ is the correct choice.
This is a neutralization reaction between sulfuric acid (a strong diprotic acid) and potassium hydroxide (a strong base). In a complete neutralization, the products are a salt (formed from the anion of the acid and the cation of the base) and water.
A) H₃O⁺ (Hydronium ion)
The hydronium ion is the species that makes a solution acidic. In a complete neutralization reaction with stoichiometric amounts of acid and base, the H⁺ from the acid and the OH⁻ from the base combine to form neutral H₂O molecules. There should be no excess H⁺ (or H₃O⁺) remaining as a primary product if the reaction goes to completion as written.
B) HSO₃⁻ (Hydrogen sulfite ion)
This is the conjugate base of sulfurous acid (H₂SO₃), not sulfuric acid (H₂SO₄). Sulfuric acid contains sulfate (SO₄²⁻), not sulfite (SO₃²⁻). This ion would not be a product of this reaction.
C) K₂SO₄ (Potassium sulfate)
This is the correct salt product. The balanced molecular equation is:
H₂SO₄(aq) + 2KOH(aq) → K₂SO₄(aq) + 2H₂O(l)
The two H⁺ ions from the sulfuric acid react with the two OH⁻ ions from the two potassium hydroxide molecules to form two water molecules. The remaining ions two K⁺ ions and one SO₄²⁻ ion combine to form the neutral ionic compound potassium sulfate.
D) KHSO₄ (Potassium hydrogen sulfate)
This compound, also known as potassium bisulfate, is an acidic salt. It forms when sulfuric acid is only partially neutralized (i.e., when one mole of KOH reacts with one mole of H₂SO₄): H₂SO₄ + KOH → KHSO₄ + H₂O. However, the reaction given shows a 1:2 mole ratio, indicating both acidic protons from sulfuric acid are neutralized. This leads to the normal sulfate salt (K₂SO₄), not the hydrogen sulfate.
Conclusion:
In a complete neutralization reaction where two moles of a monohydroxy base (KOH) react with one mole of a diprotic acid (H₂SO₄), the products are the normal salt (containing the anion with a -2 charge) and water. Therefore, the correct salt product is potassium sulfate, K₂SO₄.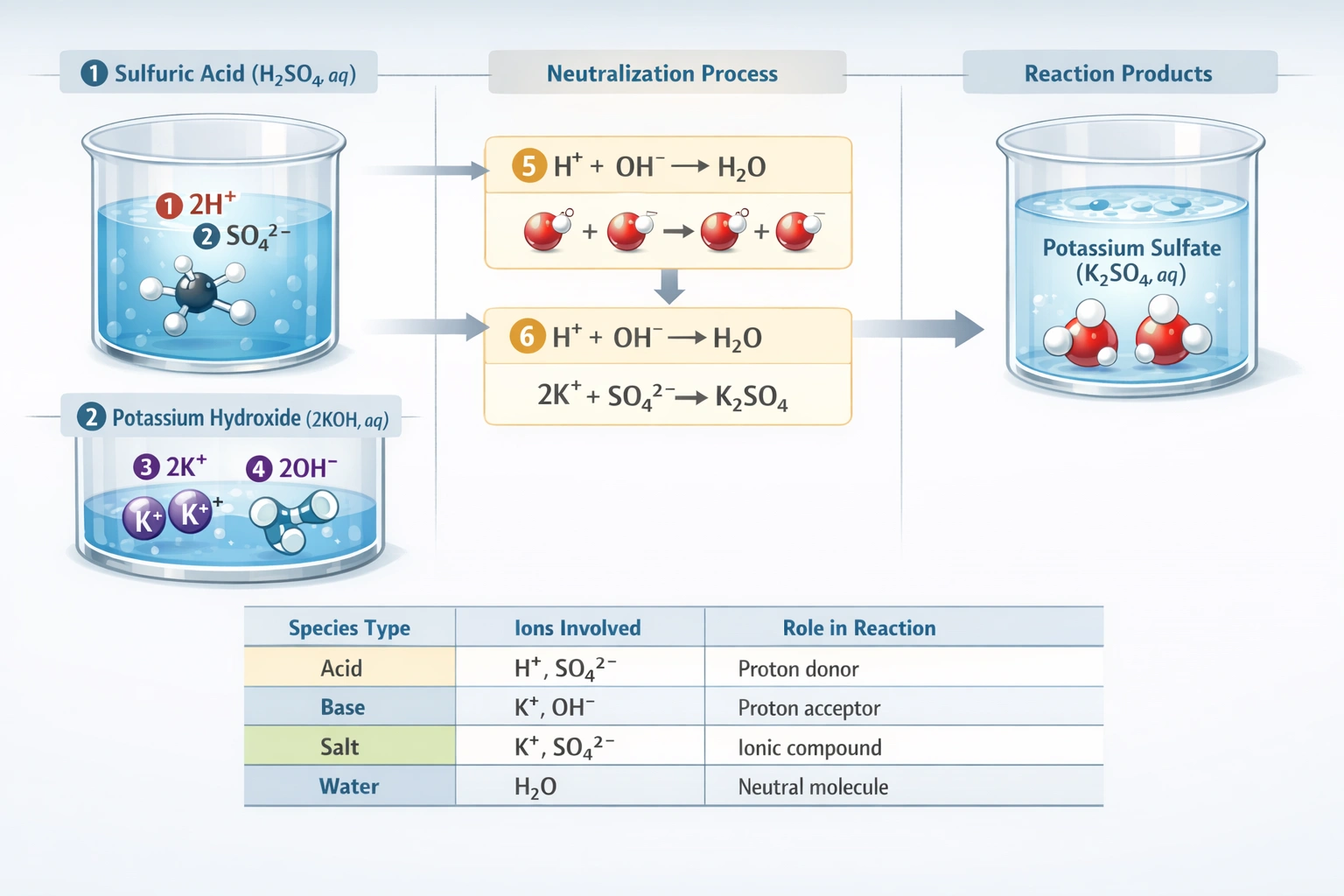
Topic Flashcards
Click to FlipWhat are the two products formed in the complete neutralization reaction between sulfuric acid (H₂SO₄) and potassium hydroxide (KOH)?
Potassium sulfate (K₂SO₄) and water (H₂O).
Write the balanced chemical equation for the complete reaction of one molecule of H₂SO₄ with KOH.
H₂SO₄ + 2KOH → K₂SO₄ + 2H₂O
In the balanced reaction H₂SO₄ + 2KOH → products, what is the name and formula of the ionic compound (salt) produced?
Potassium sulfate, K₂SO₄
What salt is produced if only one mole of KOH reacts with one mole of H₂SO₄ (incomplete neutralization)?
Potassium hydrogen sulfate (or potassium bisulfate), KHSO₄
What type of chemical reaction is H₂SO₄ + 2KOH → K₂SO₄ + 2H₂O?
An acid-base neutralization reaction (a specific type of double displacement reaction).