According to Charles’s Law, at constant pressure, Kelvin temperature is proportional to _______.
-
A
mass
-
B
volume
-
C
expansion
-
D
compression
According to Charles’s Law, at constant pressure, Kelvin temperature is proportional to volume.
Charles’s Law describes the direct relationship between the temperature of a gas and its volume when the pressure and amount of gas remain constant. As the absolute (Kelvin) temperature increases, gas particles move faster and spread farther apart, causing the volume to increase proportionally.
A) mass
Mass refers to the amount of matter in the gas and does not change with temperature in a closed system. Charles’s Law does not relate temperature to mass, so this variable is not part of the relationship.
B) volume
At constant pressure, volume increases linearly with absolute temperature. This is the core statement of Charles’s Law, often written as V∝TV \propto TV∝T (with temperature in kelvins). Heating a gas causes it to expand; cooling causes it to contract, provided pressure is held constant.
C) expansion
Expansion is a descriptive outcome rather than a measurable physical variable in the law. Charles’s Law specifically links temperature to volume, not to the qualitative idea of expansion itself.
D) compression
Compression implies a decrease in volume, which would occur when temperature decreases, not increases. This option describes the opposite behavior of what Charles’s Law predicts when temperature rises.
Conclusion
Charles’s Law states that, at constant pressure, the volume of a gas is directly proportional to its Kelvin temperature.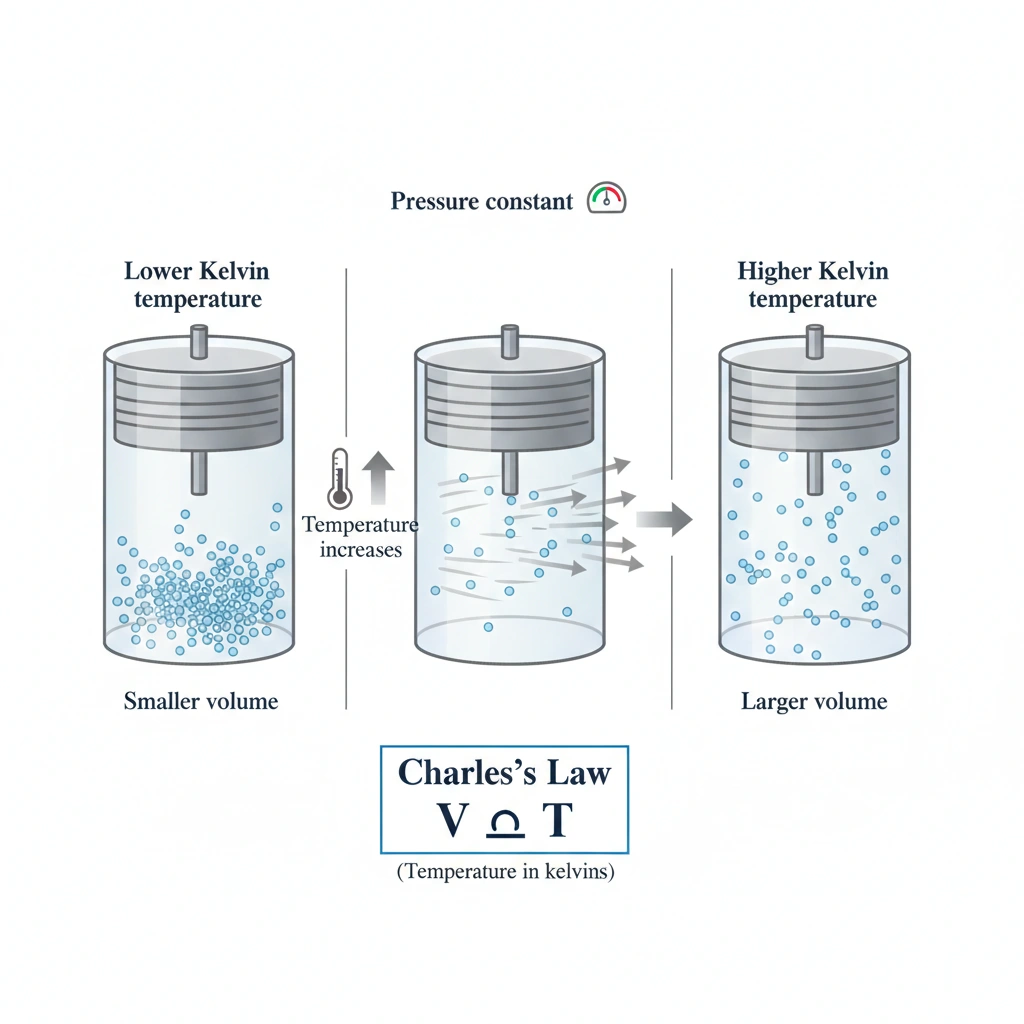
Topic Flashcards
Click to FlipState Charles's Law in your own words. What condition (which variable is held constant) is required?
At constant pressure, the volume of a fixed amount of gas is directly proportional to its absolute temperature (in Kelvin).
Write the mathematical expression for Charles's Law. What do the variables V and T represent?
V₁/T₁ = V₂/T₂, where V is volume and T is absolute temperature in Kelvin.
Why is the Kelvin temperature scale (not Celsius) essential for using Charles's Law?
The direct proportionality (V ∝ T) is only valid for an absolute temperature scale where zero represents the theoretical point of zero volume. The Kelvin scale starts at absolute zero (0 K = -273.15 °C), making it the correct scale for gas laws.
If a balloon has a volume of 2.0 L at 300 K, what will its volume be at 450 K if the pressure is constant?
Using V₁/T₁ = V₂/T₂: V₂ = V₁ * (T₂/T₁) = 2.0 L * (450 K / 300 K) = 3.0 L.
Explain Charles's Law using the concepts of kinetic molecular theory (the behavior of gas particles).
Increasing the temperature increases the average kinetic energy of the gas particles. At constant pressure, the particles move faster and collide with the container walls more forcefully, causing the walls to expand until the pressure inside again equals the constant external pressure, resulting in a larger volume.