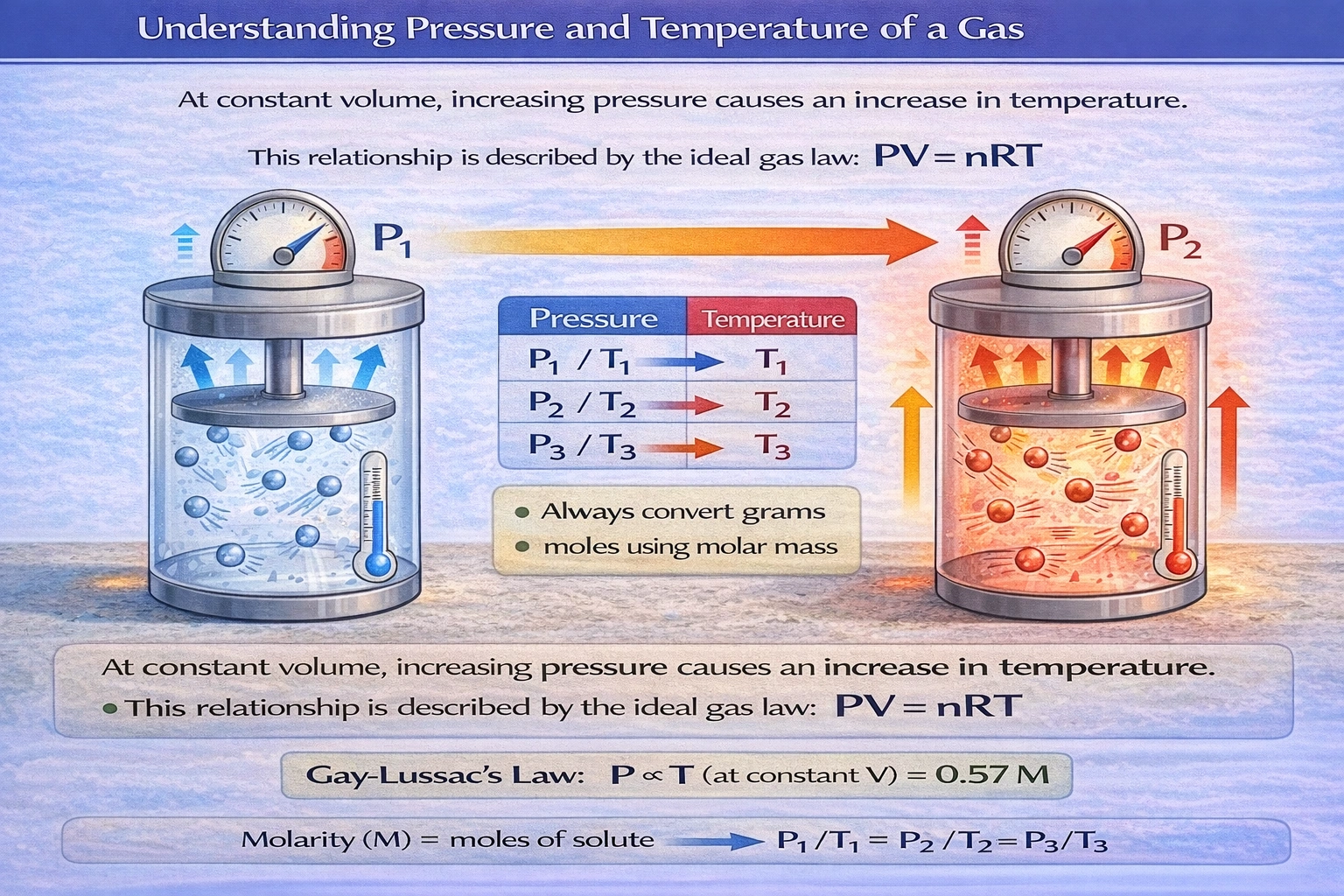
Considering a gas in a closed system, at a constant volume, what will happen to the temperature if the pressure is increased?
-
A
The temperature will stay the same
-
B
The temperature will decrease
-
C
The temperature will increase
-
D
It cannot be determined with the information given
According to Gay-Lussac's law (P ∝ T at constant volume), increasing pressure in a closed system with fixed volume necessitates a proportional temperature increase to maintain the pressure-temperature relationship described by the ideal gas law.
The ideal gas law (PV = nRT) establishes direct proportionality between pressure and temperature when volume and moles of gas remain constant—meaning pressure elevation requires corresponding temperature elevation to satisfy the equation's constraints.
A) The temperature will stay the same
Constant temperature with increased pressure at fixed volume violates the ideal gas law. If V and n are constant while P increases, T must increase proportionally to maintain PV = nRT equality. Isothermal compression (constant temperature) requires volume reduction—not applicable here since volume is constrained. Holding temperature constant while increasing pressure at fixed volume would require adding gas molecules (increasing n), but the question specifies a closed system where n remains constant.
B) The temperature will decrease
Inverse pressure-temperature relationship contradicts kinetic molecular theory. Gas pressure arises from molecular collisions with container walls; increased collision frequency/force elevates pressure. At fixed volume, only increased molecular kinetic energy (temperature) can produce greater collision force/frequency—decreased temperature would reduce pressure, not increase it. This option inverts the fundamental relationship governing gas behavior.
C) The temperature will increase
Gay-Lussac's law states P₁/T₁ = P₂/T₂ for constant volume and moles. Rearranging: T₂ = T₁ × (P₂/P₁). If P₂ > P₁, then T₂ > T₁—temperature must rise proportionally with pressure. Microscopically, increased temperature elevates average molecular kinetic energy (½mv² = 3/2kT), causing more frequent and forceful wall collisions that manifest as higher pressure. In rigid containers (fixed volume), pressure elevation can only result from temperature increase or added gas molecules—closed systems exclude the latter possibility.
D) It cannot be determined with the information given
The question provides sufficient constraints: closed system (constant n), constant volume (V fixed), and pressure increase (P rising). These three conditions uniquely determine temperature must increase according to PV = nRT. No additional information is required the relationship is mathematically determined by the ideal gas law's constraints.
Conclusion:
Gas behavior follows mathematically precise relationships governed by the ideal gas law. When two variables are constrained (V and n constant) and a third changes (P increases), the fourth variable (T) is deterministically fixed by the equation's requirements. Option C correctly applies Gay-Lussac's law to predict temperature elevation a principle exploited in pressure cookers (increased T raises P at fixed V) and observed in automobile tires (pressure rises on hot days). Understanding these constrained relationships proves essential for engineering applications, meteorological prediction, and physiological processes like pulmonary gas exchange where volume, pressure, and temperature interact dynamically.