During which of the following phase changes will the cohesion between the particles in a substance increase?
-
A
Solid to gas
-
B
Liquid to gas
-
C
Gas to liquid
-
D
Solid to liquid
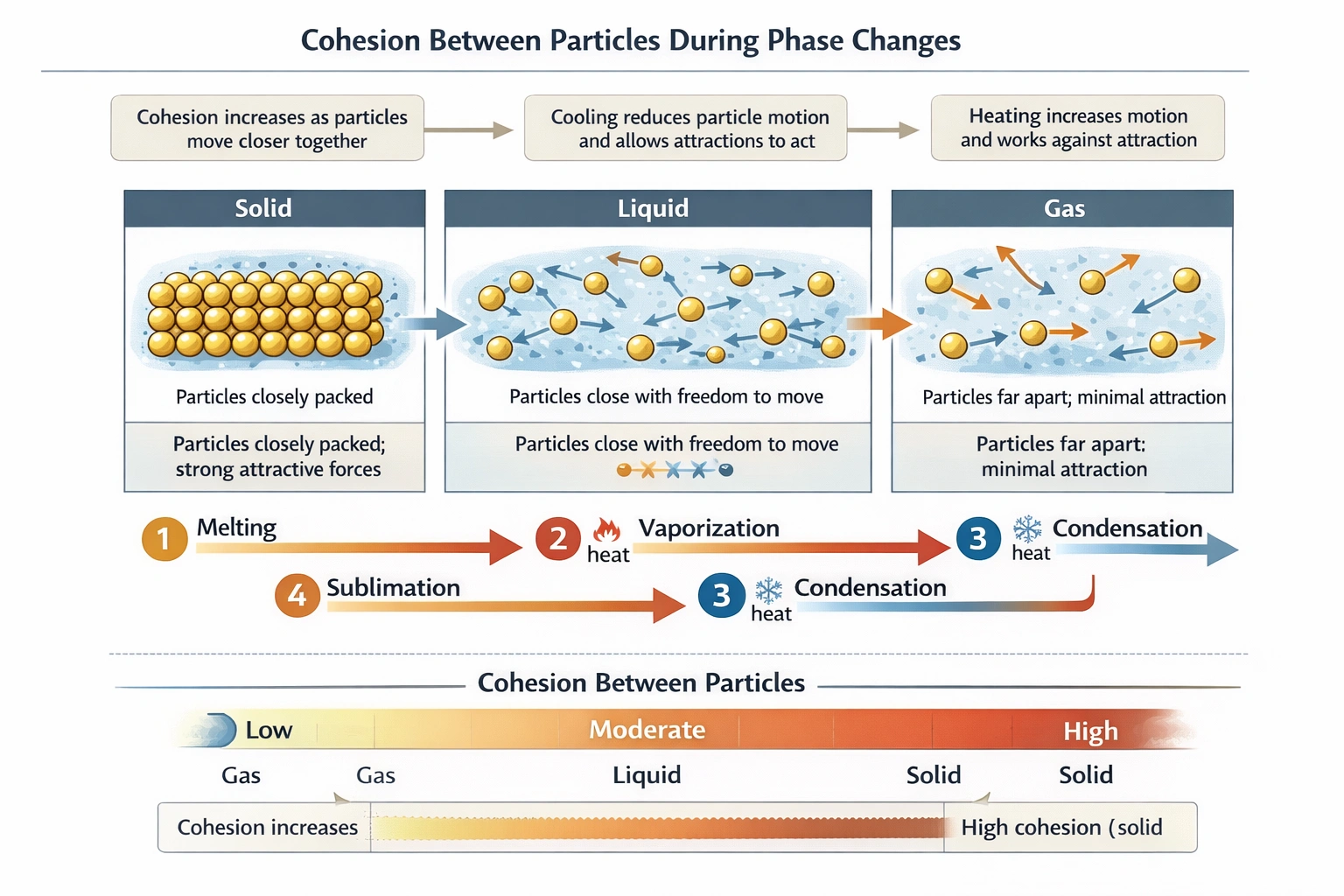
Cohesion between particles increases during the phase change from gas to liquid.
Cohesion refers to the attractive force between particles of the same substance. The strength of these cohesive forces is directly related to the proximity of the particles and the opportunity for intermolecular interactions such as hydrogen bonding, dipole-dipole forces, or London dispersion forces to act.
A) Solid to gas (Sublimation)
Sublimation requires a substantial input of energy to overcome the very strong cohesive forces holding the solid lattice together. Particles transition directly from a tightly bound, ordered solid to a widely dispersed gas where intermolecular forces are minimal. Cohesion decreases dramatically and becomes almost negligible.
B) Liquid to gas (Vaporization/Boiling)
Vaporization also requires an input of energy (the heat of vaporization) to give particles enough kinetic energy to break free from the cohesive forces of the liquid state. As particles escape into the gas phase, the average distance between them increases enormously, and the strength of cohesive interactions plummets.
C) Gas to liquid (Condensation)
Condensation is an exothermic process where a gas releases energy and cools. As particles lose kinetic energy, they slow down and come closer together. This decreased distance allows intermolecular attractive forces to become effective, pulling the particles into the more condensed liquid state. This transition from a state of minimal interaction (gas) to a state where particles are in constant close contact (liquid) represents a significant increase in cohesion.
D) Solid to liquid (Melting/Fusion)
Melting requires energy to disrupt the rigid, highly ordered structure of a solid. While particles in a liquid are still close and cohesive forces remain strong enough to give a liquid its surface tension and viscosity, the act of melting typically involves a slight increase in the average intermolecular distance as the lattice breaks down. This often results in a slight decrease in the overall cohesive energy density compared to the solid, though liquids still exhibit substantial cohesion.
 Conclusion:
Conclusion:
Cohesive forces become stronger as particles move from a dispersed state to a condensed state. The transition from the highly dispersed gas phase to the much denser liquid phase (condensation) is where the increase in particle proximity and interaction, and thus cohesion, is most pronounced.
Topic Flashcards
Click to FlipDefine "cohesion" in terms of particles within a substance.
The attractive force (intermolecular force) between particles of the same kind.
During which phase change do particles release energy, slow down, and move closer together, allowing intermolecular forces to increase?
Condensation (gas to liquid).
Why does cohesion decrease during vaporization (liquid to gas)?
Particles gain kinetic energy to overcome attractive forces and move far apart, minimizing interactions.
Rank the three common states of matter (solid, liquid, gas) in order of INCREASING cohesive forces between particles.
Gas (weakest), Liquid, Solid (strongest).
What happens to the average distance between particles during condensation, and how does this affect cohesion?
The distance decreases significantly; this allows intermolecular forces to act more effectively, increasing cohesion.