Extract:
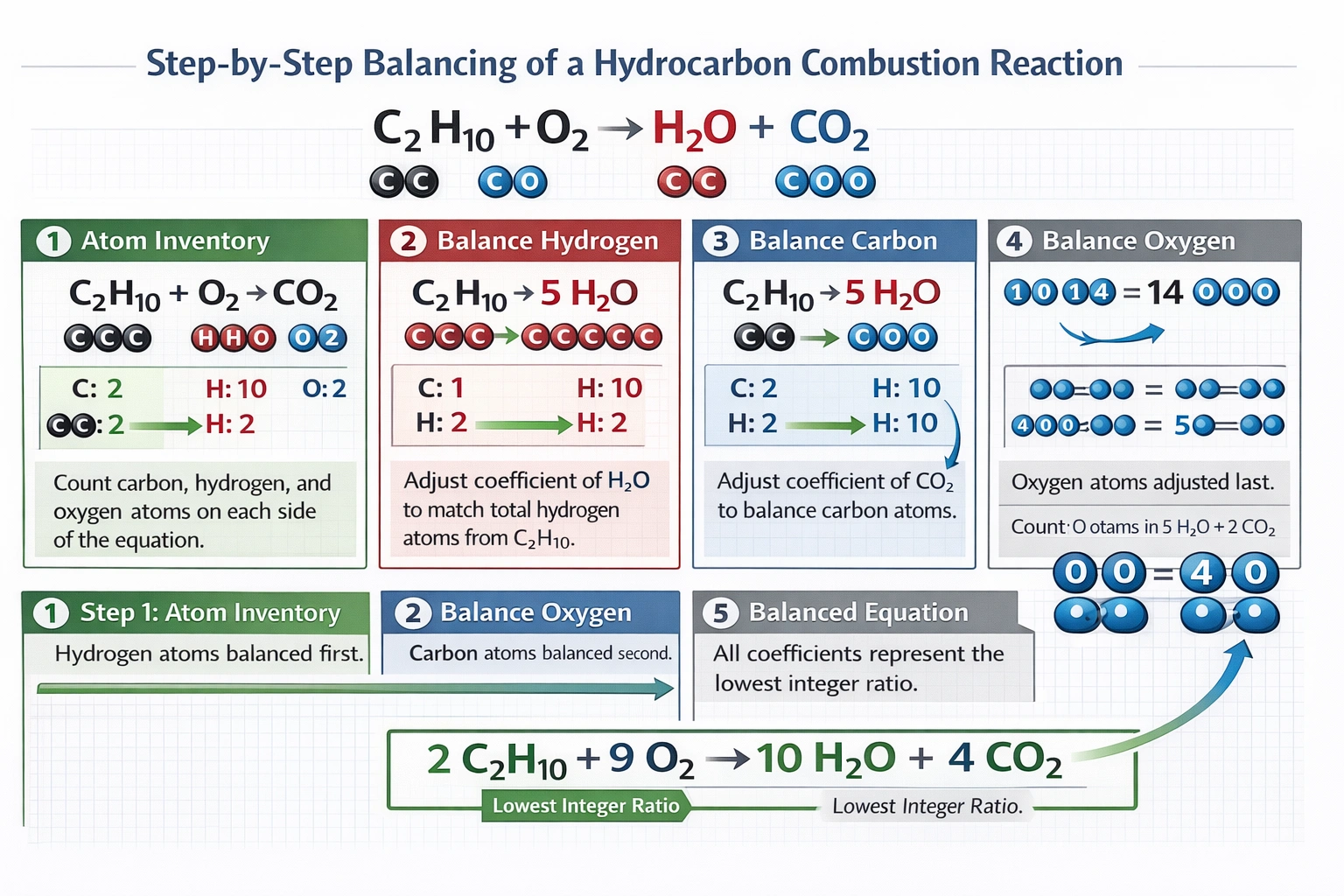
__C₂H₁₀ + __O₂ → __H₂O + __CO₂
Find the lowest coefficients that will balance the following combustion equation.
-
A
1:5:5:2
-
B
4:10:20:8
-
C
2:9:10:4
-
D
2:5:10:4
The coefficients 2:9:10:4 balance the equation 2C₂H₁₀ + 9O₂ → 10H₂O + 4CO₂, satisfying conservation of mass for carbon, hydrogen, and oxygen atoms.
Note: C₂H₁₀ is not a chemically stable hydrocarbon (ethane is C₂H₆; butane is C₄H₁₀), but the mathematical balancing proceeds based on the formula as presented in the question.
A) 1:5:5:2
Substituting coefficients yields C₂H₁₀ + 5O₂ → 5H₂O + 2CO₂. Atom inventory reveals: left side contains 2 carbon, 10 hydrogen, and 10 oxygen atoms; right side contains 2 carbon (from 2CO₂), 10 hydrogen (from 5H₂O), but 13 oxygen atoms (5 from H₂O + 8 from CO₂ = 13). Oxygen atoms are unbalanced (10 ≠ 13), violating the law of conservation of mass. This coefficient set fails to satisfy stoichiometric requirements.
B) 4:10:20:8
Substituting yields 4C₂H₁₀ + 10O₂ → 20H₂O + 8CO₂. Left side contains 8 carbon, 40 hydrogen, and 20 oxygen atoms; right side contains 8 carbon (from 8CO₂), 40 hydrogen (from 20H₂O), but 36 oxygen atoms (20 from H₂O + 16 from CO₂ = 36). Oxygen imbalance persists (20 ≠ 36). Additionally, these coefficients share a common factor of 4 and are not in lowest terms—reducing to 1:2.5:5:2 would yield non-integer coefficients, indicating this set is neither balanced nor minimal.
C) 2:9:10:4
Substituting yields 2C₂H₁₀ + 9O₂ → 10H₂O + 4CO₂. Left side atom count: carbon = 2 × 2 = 4, hydrogen = 2 × 10 = 20, oxygen = 9 × 2 = 18. Right side atom count: carbon = 4 × 1 = 4 (from 4CO₂), hydrogen = 10 × 2 = 20 (from 10H₂O), oxygen = (10 × 1) + (4 × 2) = 10 + 8 = 18 (from H₂O and CO₂ combined). All atoms balance perfectly (C: 4=4, H: 20=20, O: 18=18). These coefficients share no common divisor greater than 1, representing the lowest integer ratio that satisfies mass conservation.
D) 2:5:10:4
Substituting yields 2C₂H₁₀ + 5O₂ → 10H₂O + 4CO₂. Left side contains 4 carbon, 20 hydrogen, and 10 oxygen atoms; right side contains 4 carbon, 20 hydrogen, but 18 oxygen atoms (10 from H₂O + 8 from CO₂ = 18). Oxygen atoms remain unbalanced (10 ≠ 18), indicating insufficient oxygen molecules to fully oxidize the hydrocarbon to water and carbon dioxide. This coefficient set would imply incomplete combustion producing carbon monoxide or soot—contradicting the equation's products specifying only H₂O and CO₂.
Conclusion:
Balancing chemical equations requires equal numbers of each atom type on both sides, reflecting the law of conservation of mass. Option C provides coefficients (2, 9, 10, 4) that achieve perfect atom balance for carbon, hydrogen, and oxygen while representing the lowest integer ratio—no smaller whole numbers satisfy the equation. Although C₂H₁₀ does not represent a stable hydrocarbon (real alkanes follow CₙH₂ₙ₊₂, making C₂H₆ ethane and C₄H₁₀ butane), the mathematical solution for the given formula remains valid within the problem's constraints. Option C correctly identifies the minimal balanced coefficient set.