What is the purpose of a catalyst?
-
A
To increase a reaction's rate by increasing the activation energy
-
B
To increase a reaction's rate by increasing the temperature
-
C
To increase a reaction's rate by decreasing the activation energy
-
D
To increase a reaction's rate by decreasing the temperature
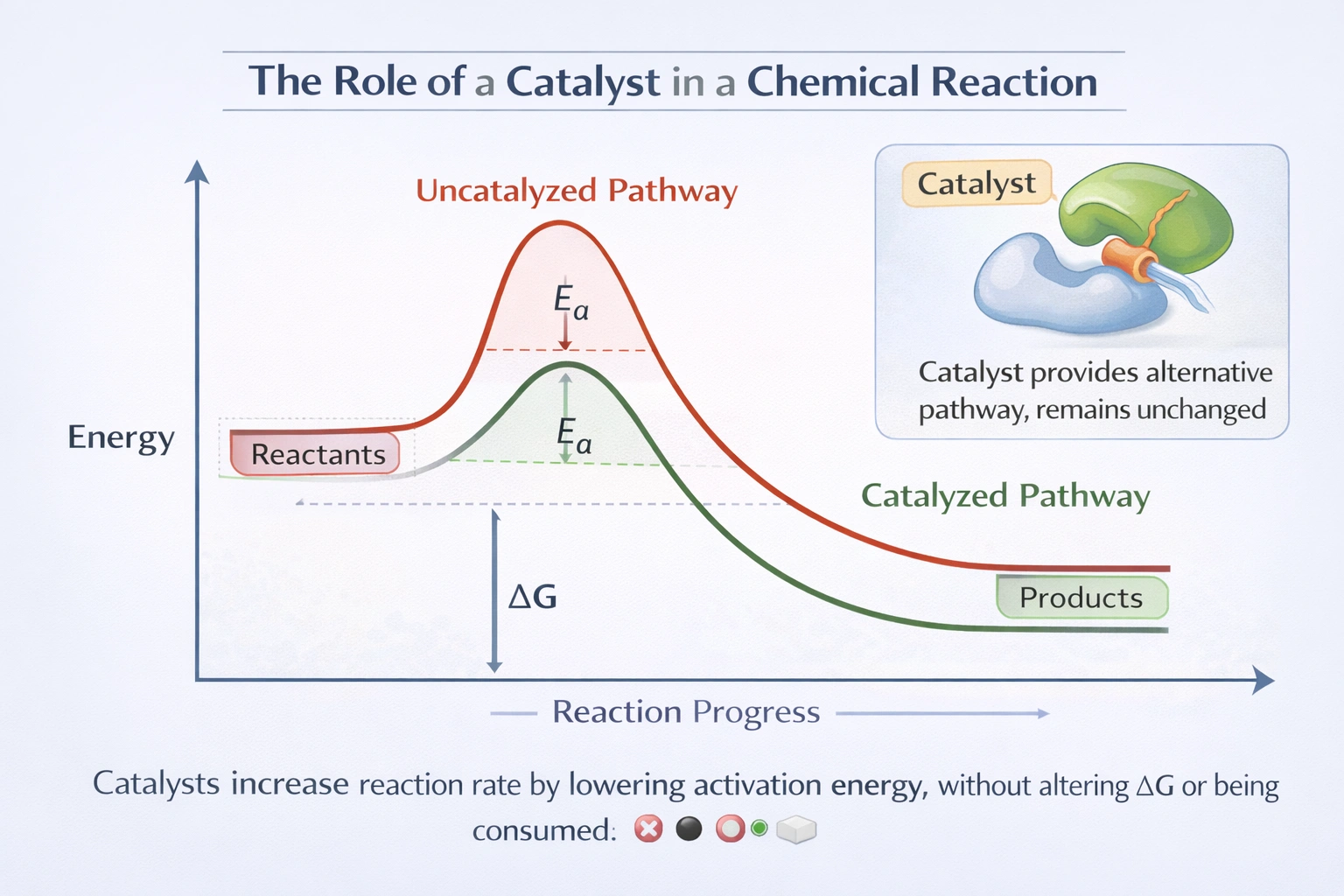
A catalyst increases a reaction's rate by providing an alternative reaction pathway with a lower activation energy barrier while remaining chemically unchanged at the reaction's completion.
Catalysts operate by stabilizing transition states or facilitating bond rearrangements through temporary interactions with reactants, enabling more molecular collisions to possess sufficient energy for successful reaction at a given temperature—without altering reaction thermodynamics (ΔG, equilibrium position) or being consumed in the process.
A) To increase a reaction's rate by increasing the activation energy
Increasing activation energy would decrease reaction rate according to the Arrhenius equation (k = Ae^(-Ea/RT)), as fewer molecules would possess sufficient kinetic energy to overcome the higher energy barrier at a given temperature. Catalysts function oppositely—they lower activation energy to increase the fraction of successful collisions. This option describes an inhibitor's effect, not a catalyst's mechanism.
B) To increase a reaction's rate by increasing the temperature
Temperature elevation increases reaction rates by providing greater kinetic energy to reactant molecules, enabling more collisions to surpass the activation energy barrier. However, catalysts operate isothermally—they accelerate reactions without requiring temperature changes. A catalyst added to a reaction at constant temperature still increases rate, demonstrating that thermal energy input is not the catalytic mechanism. Temperature modification represents an external parameter change, not an intrinsic catalytic property.
C) To increase a reaction's rate by decreasing the activation energy
Catalysts provide alternative reaction mechanisms with lower activation energy (Ea) by forming temporary enzyme-substrate complexes (in biological systems) or adsorbing reactants onto active sites (in heterogeneous catalysis). For example, catalase reduces hydrogen peroxide decomposition's Ea from approximately 75 kJ/mol to 23 kJ/mol, increasing reaction rate by a factor of 10⁹ at physiological temperatures. The catalyst stabilizes transition states through precise orientation, acid-base catalysis, or covalent intermediate formation—enabling more molecular collisions to achieve successful reaction without altering the reaction's overall free energy change or equilibrium constant.
D) To increase a reaction's rate by decreasing the temperature
Decreasing temperature reduces molecular kinetic energy, decreasing the fraction of collisions exceeding activation energy and thus slowing reaction rates according to collision theory. No catalyst operates by cooling the system; such an action would counteract rate enhancement. Catalysts function effectively at constant or even reduced temperatures compared to uncatalyzed reactions—but they do not cause the temperature decrease themselves as a mechanism of action.
Conclusion:
Catalysis fundamentally operates through transition state stabilization that lowers the activation energy barrier, enabling greater reaction velocity under identical thermal conditions. This mechanism distinguishes catalysts from other rate-affecting factors like temperature changes or concentration adjustments. Option C precisely describes this universal catalytic principle applicable across biological, industrial, and laboratory contexts—while other options confuse catalysis with inhibition, thermal effects, or thermodynamically implausible mechanisms. Catalysts remain regenerated after reaction cycles, allowing minute quantities to facilitate extensive substrate conversion without permanent chemical alteration.