Which statement is true about the pH of a solution?
-
A
A solution cannot have a pH less than 1.
-
B
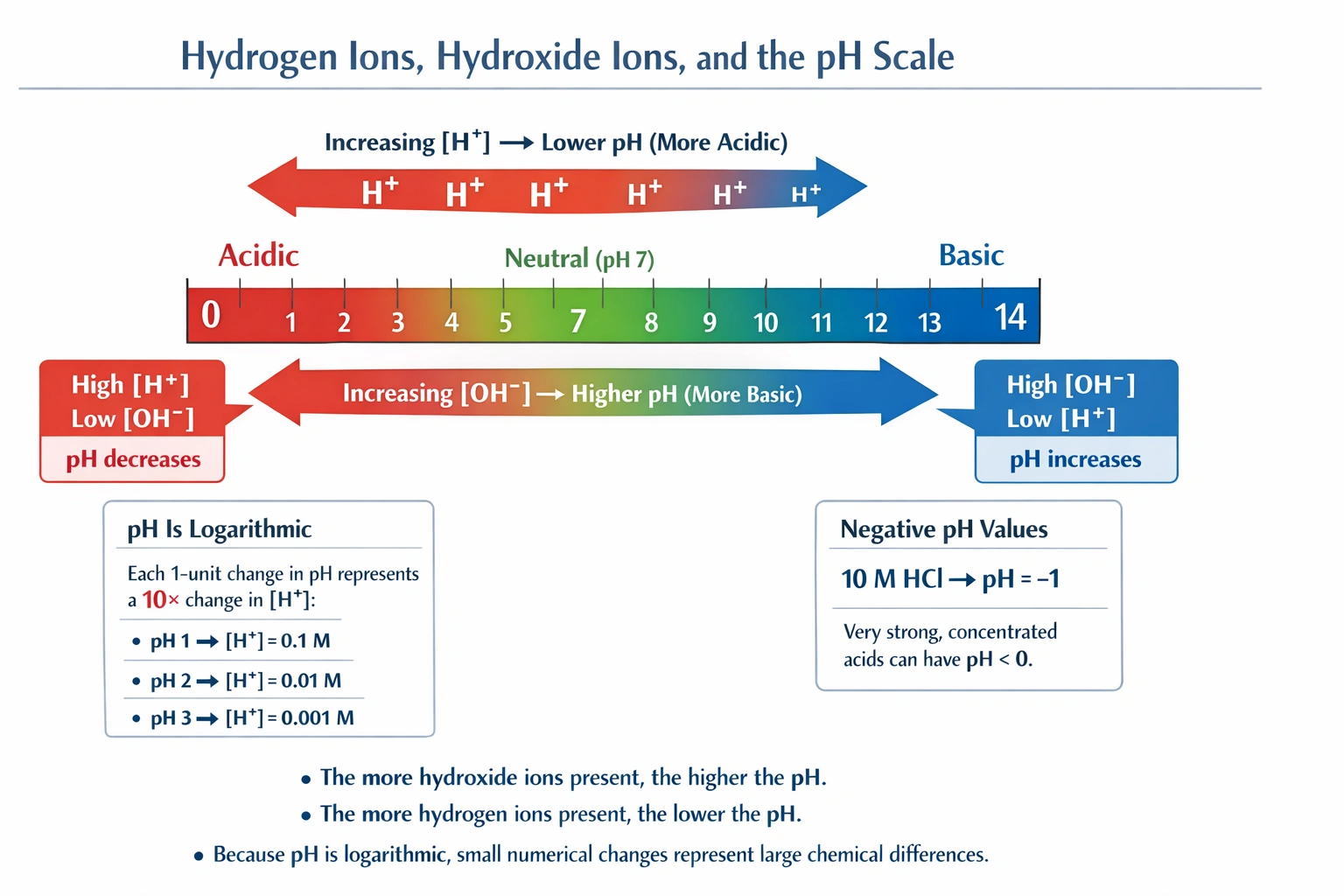
The more hydroxide ions in the solution, the higher the pH.
-
C
If an acid has a pH of greater than 2, it is considered a weak base.
-
D
A solution with a pH of 2 has ten times the amount of hydrogen ions than a solution with a power of 1.
Solutions with greater hydroxide ion concentration exhibit higher pH values because pH and pOH maintain an inverse logarithmic relationship governed by the ion product of water (Kw = [H⁺][OH⁻] = 1 × 10⁻¹⁴ at 25°C).
As hydroxide ion concentration increases, hydrogen ion concentration decreases proportionally to maintain constant Kw, resulting in elevated pH values that reflect greater alkalinity—demonstrating the fundamental reciprocal relationship between [H⁺] and [OH⁻] in aqueous systems.
A) A solution cannot have a pH less than 1
Concentrated strong acids produce pH values below 1; 10 M hydrochloric acid yields [H⁺] = 10 M, giving pH = -log(10) = -1. Similarly, 12 M HCl has pH ≈ -1.08. Negative pH values occur in highly concentrated acidic solutions and are measurable with specialized electrodes. The pH scale has no theoretical lower bound—only practical limitations based on solvent properties and acid concentration. This statement incorrectly imposes an artificial constraint on the logarithmic pH scale.
B) The more hydroxide ions in the solution, the higher the pH
The ion product of water dictates [H⁺][OH⁻] = 1 × 10⁻¹⁴ at standard temperature. As hydroxide concentration increases, hydrogen ion concentration must decrease proportionally: [H⁺] = 10⁻¹⁴/[OH⁻]. Since pH = -log[H⁺], decreasing [H⁺] produces higher pH values. A solution with [OH⁻] = 10⁻² M has [H⁺] = 10⁻¹² M and pH = 12, while [OH⁻] = 10⁻⁴ M gives [H⁺] = 10⁻¹⁰ M and pH = 10—demonstrating the direct relationship between hydroxide concentration and pH elevation. This principle underlies base strength classification and buffer behavior.
C) If an acid has a pH of greater than 2, it is considered a weak base
Acidity and basicity are defined by a substance's behavior in solution, not arbitrary pH thresholds. Acids—whether strong (HCl) or weak (acetic acid)—produce pH < 7 in aqueous solution. A weak acid at low concentration may have pH > 2 (e.g., 0.001 M acetic acid has pH ≈ 3.9) but remains chemically an acid that donates protons, not a base. Basicity requires hydroxide donation or proton acceptance capacity. pH alone cannot reclassify a chemical's fundamental nature; concentration and dissociation constant (Ka) jointly determine solution pH for acids.
D) A solution with a pH of 2 has ten times the amount of hydrogen ions than a solution with a power of 1
pH represents a logarithmic scale where each unit decrease corresponds to a tenfold hydrogen ion concentration increase. A pH 1 solution has [H⁺] = 10⁻¹ = 0.1 M, while pH 2 has [H⁺] = 10⁻² = 0.01 M—meaning pH 1 contains ten times more H⁺ than pH 2, not the reverse. The statement reverses the relationship and contains ambiguous phrasing ("power of 1" likely intends "pH of 1"). Correctly stated: a solution with pH 1 has ten times the hydrogen ion concentration of a solution with pH 2.
Conclusion:
pH functions as a logarithmic measure of hydrogen ion activity where [H⁺] = 10^(-pH), creating an inverse relationship with hydroxide concentration through water's autoionization equilibrium. Option B correctly describes how increasing hydroxide concentration elevates pH—a consequence of the fixed ion product Kw that governs all aqueous acid-base chemistry. Other options fail due to scale misconceptions (A), chemical classification errors (C), or inverted logarithmic relationships (D). Understanding this hydroxide-pH correlation proves essential for predicting buffer behavior, titration curves, and physiological acid-base balance.