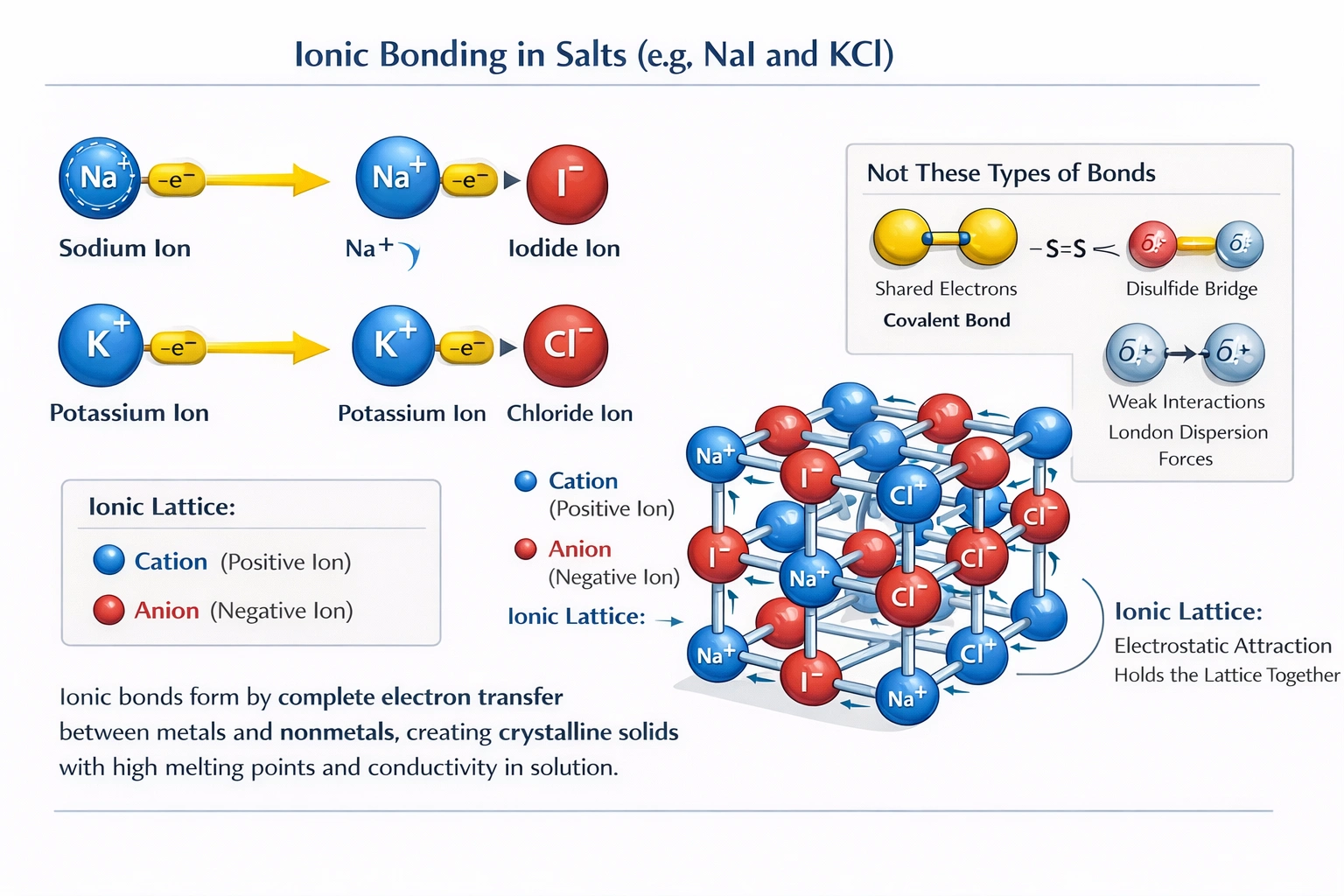
Salts like sodium iodide (NaI) and potassium chloride (KCl) use what type of bond?
-
A
Ionic bonds
-
B
Disulfide bridges
-
C
Covalent bonds
-
D
London dispersion forces
Sodium iodide and potassium chloride form ionic bonds.
These compounds exemplify classic ionic bonding where electropositive alkali metals (Na, K) donate valence electrons to electronegative halogens (I, Cl), generating Na⁺/I⁻ and K⁺/Cl⁻ ion pairs that assemble into extended three-dimensional crystal structures stabilized by non-directional Coulombic forces between oppositely charged ions.
A) Ionic bonds
Ionic bonding occurs between atoms with large electronegativity differences (>1.7 on Pauling scale), typically metals and nonmetals. Sodium (EN = 0.93) transfers its 3s¹ electron to iodine (EN = 2.66), forming Na⁺ and I⁻ ions; potassium (EN = 0.82) transfers its 4s¹ electron to chlorine (EN = 3.16), forming K⁺ and Cl⁻ ions. The resulting electrostatic attraction creates high-melting-point crystalline solids (NaI mp = 661°C; KCl mp = 770°C) that dissolve in polar solvents to yield conductive electrolyte solutions. X-ray crystallography reveals face-centered cubic lattices where each cation coordinates with six anions and vice versa—characteristic of ionic crystal packing.
B) Disulfide bridges
Disulfide bridges (S-S bonds) are covalent linkages formed between cysteine thiol groups in proteins, stabilizing tertiary structure (e.g., in antibodies or insulin). These bonds require sulfur-containing amino acids and reducing/oxidizing environments—conditions absent in simple inorganic salts like NaI or KCl, which contain no sulfur atoms and lack molecular structures capable of forming such bridges. Disulfide bonds represent specialized covalent interactions in biochemistry, not general salt bonding mechanisms.
C) Covalent bonds
Covalent bonds involve electron sharing between atoms with similar electronegativities (difference <1.7), creating discrete molecules with directional bonds (e.g., H₂O, CH₄). NaI and KCl exhibit no electron sharing; their complete electron transfer produces ions that lack molecular identity in the solid state. Evidence against covalency includes high solubility in water with dissociation into free ions (conductivity measurements), absence of discrete NaI or KCl molecules in vapor phase at moderate temperatures, and crystal structures showing non-directional ion packing rather than fixed bond angles characteristic of covalent networks.
D) London dispersion forces
London dispersion forces are weak, transient attractive forces arising from instantaneous dipoles in nonpolar molecules or atoms (e.g., between noble gas atoms or hydrocarbon chains). These forces operate in all matter but dominate only in nonpolar substances with low boiling points (e.g., methane bp = -161°C). NaI and KCl possess high melting points and form rigid crystals—properties inconsistent with weak dispersion forces. While dispersion forces exist between ions as minor contributors, the dominant intermolecular force in ionic crystals is electrostatic attraction orders of magnitude stronger than London forces.
Conclusion:
Ionic bonding defines the structural and physical properties of alkali halide salts through electrostatic attraction between cations and anions formed by complete electron transfer. NaI and KCl exemplify this bonding type with characteristic high melting points, crystalline structures, aqueous dissociation, and conductivity—properties irreconcilable with covalent bonding, disulfide bridges, or weak dispersion forces. Option A correctly identifies ionic bonds as the fundamental interaction governing these compounds' behavior, reflecting a cornerstone principle of inorganic chemistry essential for predicting solubility, reactivity, and material properties.