Half life 10 y. Fraction left after 30 y?
-
A
½
-
B
⅓
-
C
¼
-
D
⅛
The fraction of the substance left after 30 years is one eighth.
Half-life is the time required for half of a radioactive substance to decay. The decay process is exponential, meaning the remaining amount is reduced by half during each half-life period.
In this problem, the half-life is 10 years. After 30 years, three half-lives have passed:
10 years → first half-life
20 years → second half-life
30 years → third half-life
After each half-life, the remaining fraction is multiplied by one half.
A) ½
This fraction corresponds to only one half-life having passed, which is 10 years. Since 30 years have elapsed, more decay has occurred.
B) ⅓
Radioactive decay does not follow a linear or fractional pattern like thirds. It follows exponential decay based on halves, so this value is not applicable.
C) ¼
This fraction represents two half-lives, which corresponds to 20 years. After 30 years, an additional half-life has passed, so even less remains.
D) ⅛
After three half-lives, the remaining fraction is found by multiplying one half by itself three times, which gives one eighth. This correctly represents the amount of substance left after 30 years.
Conclusion
With a half-life of 10 years, 30 years corresponds to three half-lives. After three half-lives, one eighth of the original substance remains.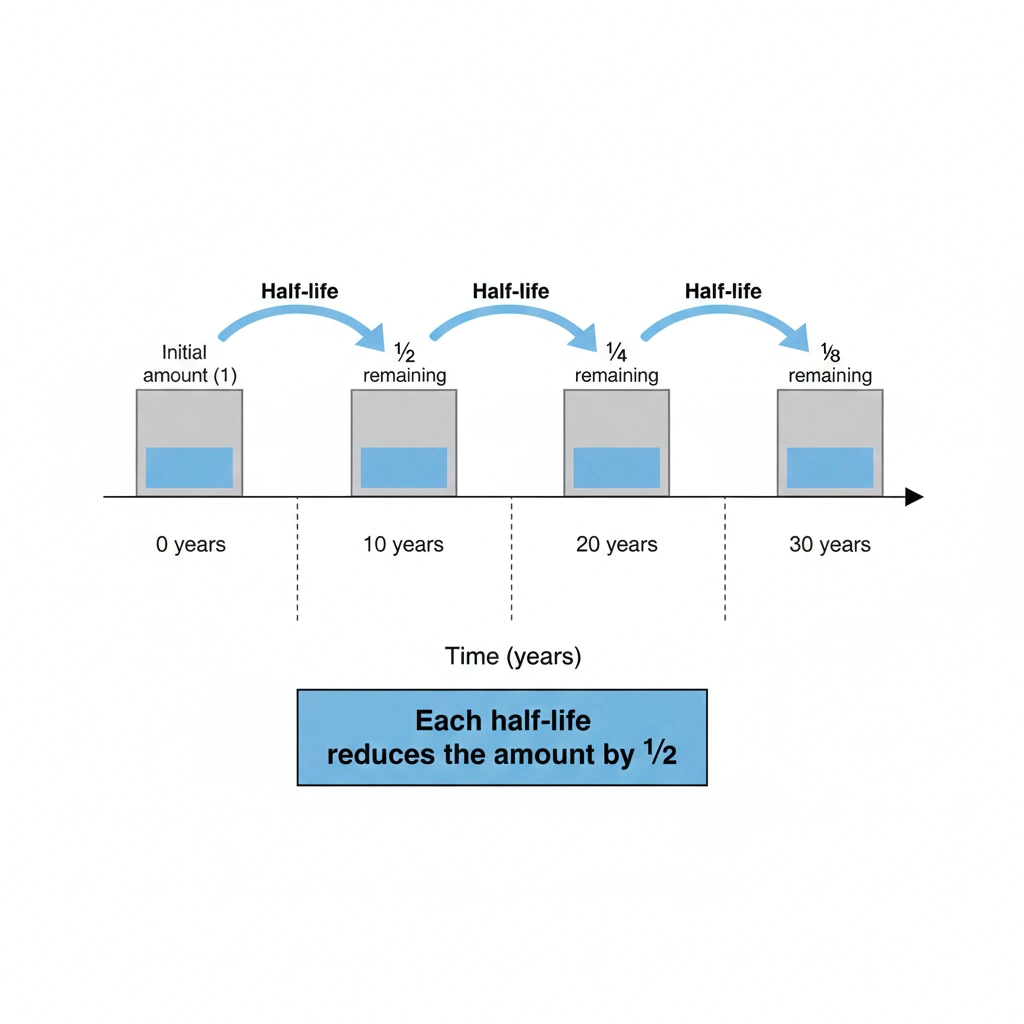
Topic Flashcards
Click to FlipWhat is the mathematical relationship between the number of half-lives (n) that have passed and the fraction of a radioactive sample remaining?
Fraction Remaining = (1/2)^n, where *n* is the number of half-lives (n = total time / half-life).
A sample has a half-life of 10 years. How much time is required for the sample to decay to 1/16 of its original amount?
40 years. 1/16 = (1/2)^4, so 4 half-lives are needed. 4 half-lives * 10 years/half-life = 40 years
If 1/8 of a sample remains after 30 years, what is its half-life? What is its decay constant (λ)?
Half-life = 10 years (3 half-lives in 30 years). Decay constant, λ = ln(2) / T_{1/2} ≈ 0.693 / 10 yr ≈ 0.0693 per year.
On a graph plotting the remaining number of atoms (N) versus time (t) for a radioactive substance, what is the shape of the curve? What does the slope at any point represent?
It is a downward-sloping exponential curve. The slope at any point represents the activity (or decay rate), which is -λN.
Why is the concept of half-life more practical for understanding radioactive decay than the decay constant (λ) for most people?
Half-life provides an intuitive, time-based measure (e.g., "half is gone in 10 years") rather than an abstract instantaneous probability of decay per unit time (λ).