Heat uses only conduction through _______.
-
A
Gases
-
B
Liquids
-
C
Solids
-
D
Solids and liquids
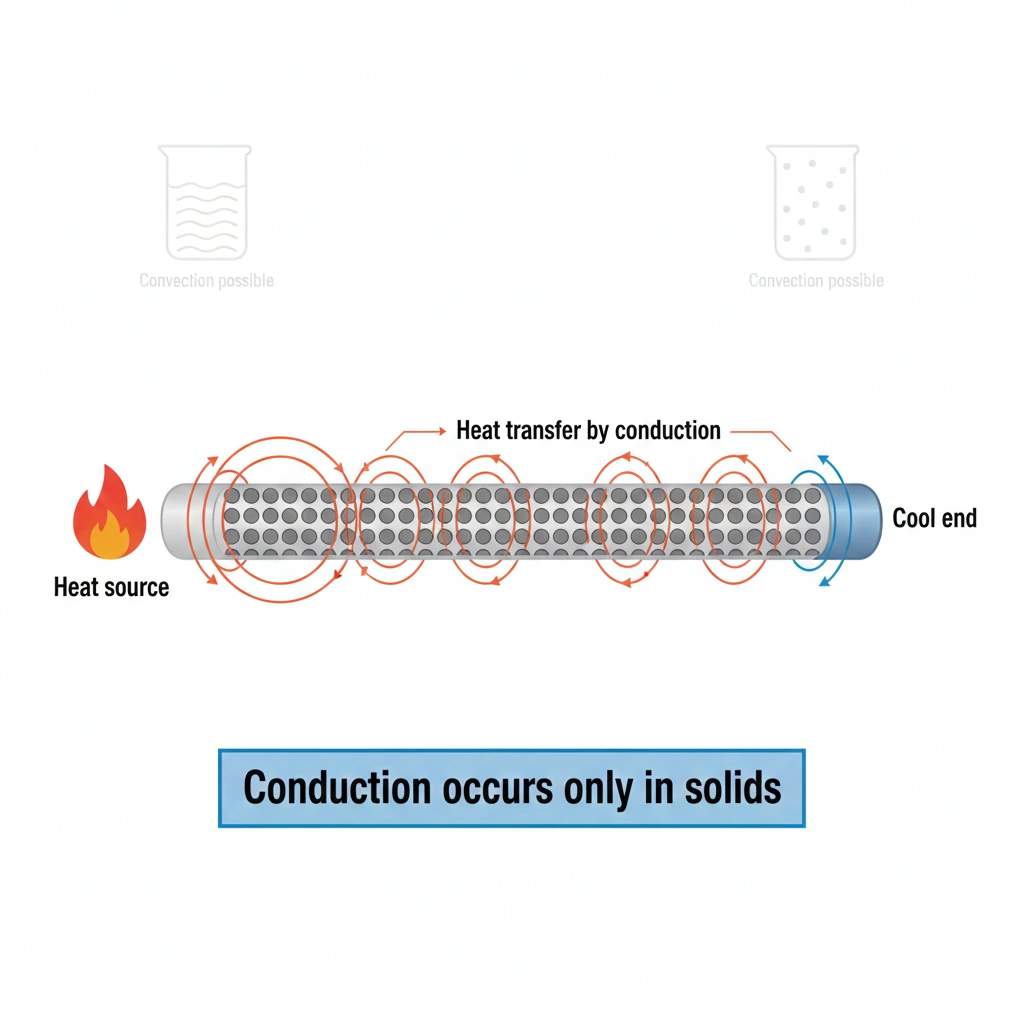
Heat is transferred only by conduction through solids.
Heat transfer can occur by conduction, convection, or radiation. Conduction involves the direct transfer of thermal energy through collisions between particles or through free electrons, without any bulk movement of the material. In solids, particles are fixed in position and cannot flow, so heat transfer occurs exclusively by conduction.
A) Gases
In gases, particles are far apart and move freely. While conduction can occur to a small extent through molecular collisions, heat transfer in gases primarily happens by convection, where warmer, less dense gas rises and cooler gas sinks. Therefore, conduction is not the only heat transfer mechanism in gases.
B) Liquids
Liquids allow particle movement, which enables convection currents to form. As a result, although conduction does occur in liquids, convection is usually the dominant mechanism for heat transfer. This means conduction is not the sole method of heat transfer in liquids.
C) Solids
In solids, particles vibrate about fixed positions and cannot move freely from place to place. Because there is no bulk motion of matter, convection cannot occur. Heat is transferred only by conduction, through lattice vibrations and, in metals, through the movement of free electrons. This makes conduction the exclusive heat transfer mechanism in solids.
D) Solids and liquids
This option is incorrect because liquids can transfer heat by convection as well as conduction. Only solids rely exclusively on conduction for heat transfer.
Conclusion
Conduction is the only mechanism of heat transfer in solids because their particles are fixed in place and cannot support convection. Therefore, heat uses only conduction through solids.
Topic Flashcards
Click to FlipWhat is the defining characteristic of conduction as a method of heat transfer, and what are the primary particles involved in solids?
Conduction is the transfer of heat through direct particle collisions and interactions without bulk movement of the material. In solids, this occurs via vibrating atoms/molecules and, in metals, mobile electrons.
Why is convection not possible in a solid block of material?
Convection requires the bulk movement of a fluid (liquid or gas) to carry heat. The particles in a solid are locked in a fixed position in a lattice and cannot flow, so convection cannot occur.
How do the three modes of heat transfer (conduction, convection, radiation) differ in terms of their requirement for a medium?
Conduction & Convection require a material medium. Radiation does not; it can travel through a vacuum (e.g., heat from the Sun).
A metal spoon and a wooden spoon are placed in a hot soup. The metal handle becomes hot quickly, while the wood stays cool longer. Both are solids, so why the difference?
The difference is due to thermal conductivity. Metals are good conductors because of their free electrons, which transfer kinetic energy rapidly. Wood is a poor conductor (good insulator)
Describe a simple real-world example where heat transfer in a solid relies only on conduction.
Heating one end of a metal rod over a flame. The other end eventually gets hot because heat travels through the rod via conduction (vibrating atoms and free electrons), with no material flowing.