If barium metal (Ba) and aluminum chloride (AlCl₃) react in a single-replacement reaction, what product(s) form?
-
A
BaAlCl₅
-
B
AlBaCl₄
-
C
Al + BaCl₂
-
D
AlCl₅ + Ba
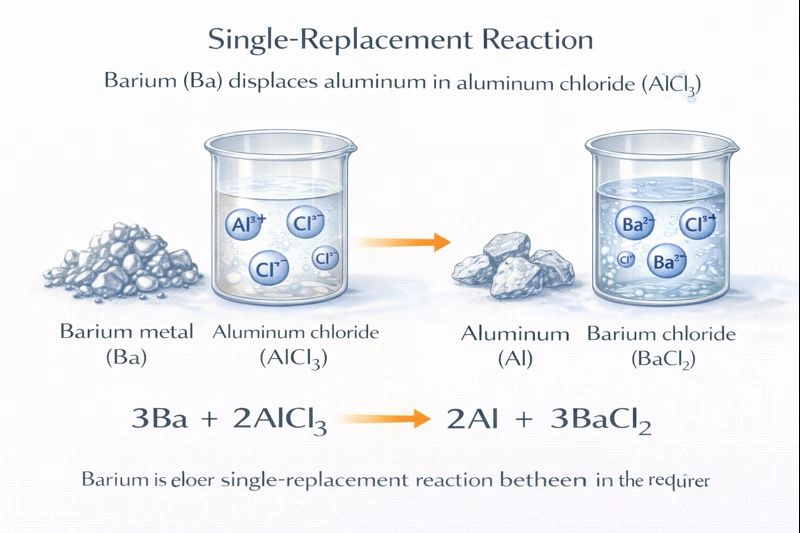
The products of this single-replacement reaction are aluminum (Al) and barium chloride (BaCl₂).
In a single-replacement (or displacement) reaction, a more reactive element (metal) displaces a less reactive one from a compound. Reactivity is determined by the activity series of metals. Barium is an alkaline earth metal and is more reactive than aluminum. Therefore, barium will displace aluminum from aluminum chloride. The barium atoms take the place of aluminum ions, forming barium chloride, and aluminum is released as a free metal. The balanced equation is: 3Ba + 2AlCl₃ → 2Al + 3BaCl₂.
A) BaAlCl₅
This suggests a single, combined product, which is characteristic of a synthesis or combination reaction, not a single-replacement reaction. Single-replacement reactions always yield an element and a new compound. Furthermore, the formula BaAlCl₅ is not a known, stable compound under normal conditions.
B) AlBaCl₄
This also suggests a synthesis into a single, complex compound. It does not represent the exchange of components required in a displacement reaction. The notation implies an alloy or mixed salt that is not the typical outcome of this type of reaction.
C) Al + BaCl₂
This correctly shows the products of the displacement. Barium replaces aluminum, resulting in elemental aluminum and the new compound barium chloride. This aligns perfectly with the pattern of a single-replacement reaction: A + BC → AC + B, where A is a more active metal than B.
D) AlCl₅ + Ba
This implies that aluminum forms a pentachloride (AlCl₅), which is not a stable compound (aluminum typically forms Al³⁺ and has a +3 oxidation state, bonding with three chloride ions to form AlCl₃). It also incorrectly shows the original aluminum chloride as a product, suggesting no reaction took place.
Conclusion:
The outcome of a single-replacement reaction is predictable using the activity series. Since barium is more reactive than aluminum, it will displace aluminum from its compound. The products are the displaced metal (aluminum) and the new compound formed by the reactive metal and the anion (barium chloride).
Topic Flashcards
Click to FlipIn a single-replacement reaction, what is the name for the element that must be more reactive in order to displace another from a compound?
The more reactive (or active) metal.
Using the activity series, what are the two products when solid barium reacts with aqueous aluminum chloride?
Aluminum metal (Al) and barium chloride (BaCl₂).
Write the balanced chemical equation for the single-replacement reaction between solid barium and aqueous aluminum chloride.
3Ba (s) + 2AlCl₃ (aq) → 2Al (s) + 3BaCl₂ (aq).
Why is a compound like "BaAlCl₅" an unlikely product in a reaction between Ba and AlCl₃?
Single-replacement reactions produce an element and a new ionic compound, not a single, complex combined salt.
hat piece of information (like a chart or list) must you consult to predict whether barium will replace aluminum in a compound?
The activity series (or reactivity series) of metals.