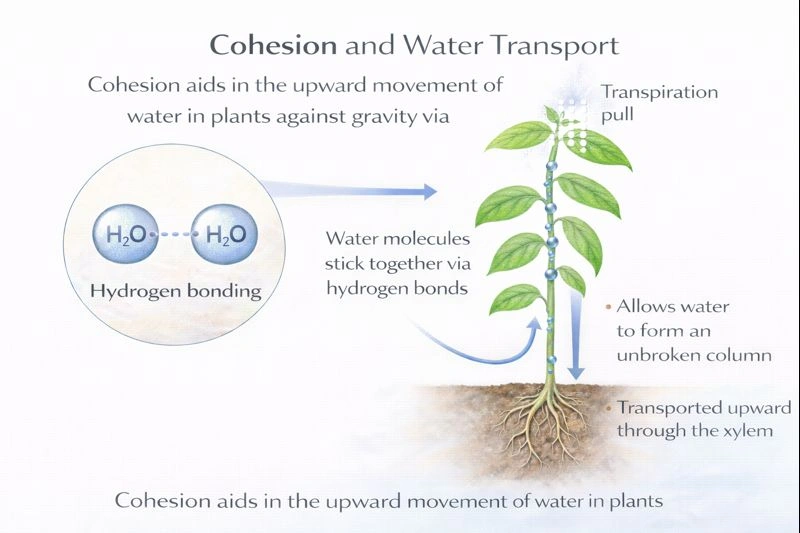
Inside a plant, water has to travel up, against gravity, to reach all the leaves. What property does this illustrate?
-
A
Adhesion
-
B
Bonding
-
C
Cohesion
-
D
Polarity
The upward movement of water in plants primarily illustrates the property of cohesion.
The transport of water from roots to leaves is driven by transpiration pull and relies on the physical properties of water. Cohesion refers to the attraction between like molecules—in this case, water molecules to other water molecules via hydrogen bonding. This cohesive force allows water to form a continuous, unbroken column within the narrow xylem vessels. As water evaporates from the leaves (transpiration), it creates a negative pressure or tension that pulls the entire water column upward. Cohesion ensures this pull is transmitted all the way down to the roots without the column breaking.
A) Adhesion
Adhesion is the attraction between water molecules and other, different substances, such as the hydrophilic walls of the xylem cells. Adhesive forces help hold the water column against the sides of the xylem and contribute to capillary action. While important, adhesion alone cannot explain how water is pulled as a continuous column against gravity over great heights; that requires the tensile strength provided by cohesion.
B) Bonding
This term is too broad and non-specific. "Bonding" can refer to intramolecular covalent bonds holding the H₂O molecule together or the intermolecular hydrogen bonds responsible for cohesion and adhesion. The specific biological term for the phenomenon described in the question is cohesion, not the general concept of bonding.
C) Cohesion
Cohesion, the mutual attraction between water molecules, is the key property that allows water to be pulled upward under tension. The hydrogen bonds between water molecules give the water column high tensile strength, enabling it to withstand the negative pressure created by transpiration without cavitation (forming air bubbles). This cohesive pull is the principal mechanism for water ascent in tall plants.
D) Polarity
Polarity is the underlying molecular cause of water's cohesive and adhesive properties. The polar nature of the water molecule, with partial positive and negative charges, enables the formation of hydrogen bonds. While polarity is the reason why cohesion exists, it is cohesion itself—the effect of those hydrogen bonds—that is the property directly illustrated by the unbroken water column moving against gravity.
Conclusion:
The movement of water in plants is a complex process involving transpiration, cohesion, and adhesion. While adhesion helps water interact with xylem walls, the critical property that allows water to be pulled upward as a continuous column, overcoming gravity and frictional forces, is the cohesion between individual water molecules. This cohesive strength is essential for water transport in vascular plants.