Name the reaction: 8Fe + S₈ → 8FeS
-
A
Single displacement
-
B
Double displacement
-
C
Synthesis
-
D
Acid-base
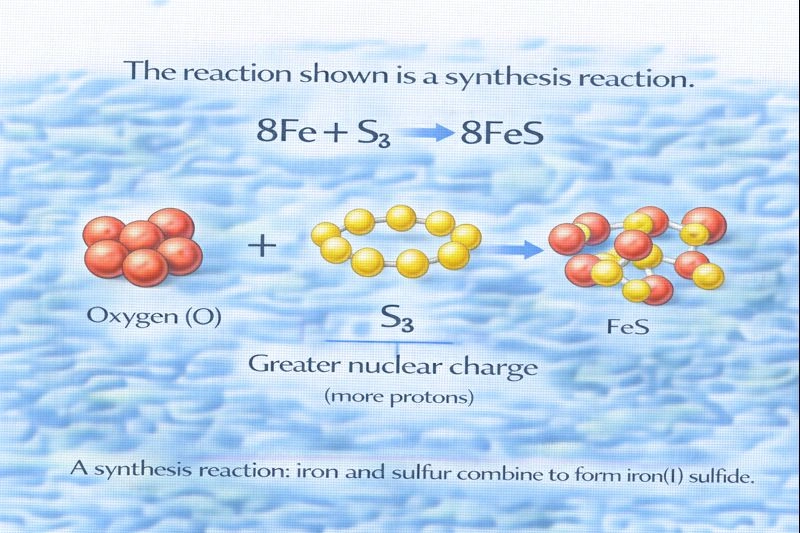
The reaction shown is a synthesis reaction.
A synthesis reaction, also known as a combination reaction, occurs when two or more simpler substances—such as elements or simple compounds—combine to form a single, more complex compound. In the given reaction, elemental iron (Fe) reacts directly with elemental sulphur (S₈) to produce iron(II) sulfide (FeS). This direct combination of elements into one compound is the defining characteristic of a synthesis reaction.
A. Single displacement
A single displacement (or single replacement) reaction involves one element replacing another element within a compound, following the general pattern A + BC → AC + B. In the given equation, there is no compound present at the start of the reaction and no element being displaced. Instead, two elements are combining, so this reaction does not fit the single displacement pattern.
B. Double displacement
A double displacement reaction involves two ionic compounds exchanging ions, typically following the pattern AB + CD → AD + CB. These reactions usually occur in aqueous solutions and involve compounds, not free elements. Since the reaction shown involves only elements reacting to form a compound, it cannot be classified as a double displacement reaction.
C. Synthesis
In a synthesis reaction, multiple reactants combine to form a single product. In this case, iron atoms react with sulphur molecules to form iron(II) sulfide. The equation is balanced to reflect that one S₈ molecule, which contains eight sulphur atoms, reacts with eight iron atoms to produce eight units of FeS. This direct formation of a compound from its elements is a textbook example of a synthesis reaction.
D. Acid-base
Acid–base reactions involve the transfer of protons (H⁺) between an acid and a base, typically resulting in the formation of a salt and water. The reaction shown does not include acids, bases, hydrogen ions, or water, so it does not meet the criteria for an acid–base reaction.
Conclusion
When two elements combine directly to form a single compound, the reaction is classified as a synthesis reaction. The combination of iron and sulphur to form iron(II) sulfide clearly follows this pattern, making synthesis the correct classification.
Topic Flashcards
Click to FlipWhat is the defining characteristic of a synthesis (combination) reaction? Give the general form.
Two or more substances (usually elements or simple compounds) combine to form one, more complex product. General form: A + B → AB.
For the reaction 2Mg + O₂ → 2MgO, explain why it is a synthesis reaction and identify a key feature of its reactants.
Two elements (Mg and O₂) combine to form a single compound (MgO). A key feature is that the reactants are in their elemental forms.
Why is the reaction 8Fe + S₈ → 8FeS not classified as single displacement? What is missing from the reactants?
Single displacement requires an element and a compound (A + BC → AC + B). Here, the reactants are only elements (Fe and S₈), with no compound for one element to displace another from.
What are the four main types of chemical reactions you would use to classify a given equation, and what is a quick way to identify a synthesis reaction from this list?
Synthesis, Decomposition, Single Displacement, Double Displacement. A quick identifier for synthesis is: Multiple reactants, one product.
Provide two real-world examples of synthesis reactions, one involving metals and one involving nonmetals.
1) Rusting: 4Fe + 3O₂ → 2Fe₂O₃. 2) Formation of ammonia (Haber process): N₂ + 3H₂ → 2NH₃.