Which is a triatomic allotrope of oxygen?
-
A
Ozone
-
B
Water
-
C
Acidic oxide
-
D
Carbon dioxide
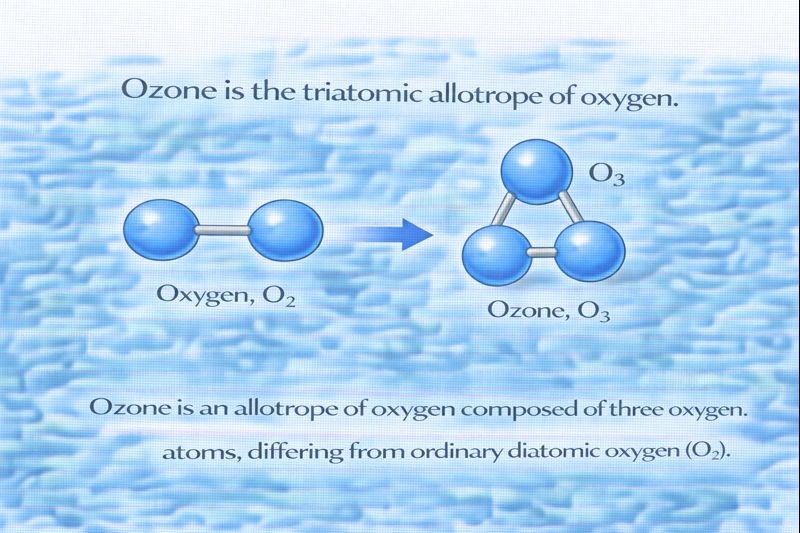
The triatomic allotrope of oxygen is ozone.
Allotropes are different structural forms of the same element that exist in the same physical state. In the case of oxygen, allotropes are made up entirely of oxygen atoms but differ in how those atoms are bonded together. The most common form of oxygen in the atmosphere is diatomic oxygen (O₂), but oxygen can also exist in a triatomic form, where three oxygen atoms are bonded together. This triatomic form is known as ozone (O₃).
A. Ozone
Ozone is a molecule composed of three oxygen atoms (O₃), making it triatomic. Because it consists solely of oxygen atoms arranged in a different molecular structure than O₂, it qualifies as an allotrope of oxygen. Ozone has distinct chemical and physical properties compared to diatomic oxygen, such as a sharp odor and a strong oxidizing ability, which arise from its unique triatomic structure.
B. Water
Water (H₂O) is a compound made of hydrogen and oxygen atoms chemically bonded together. Although it contains oxygen, it is not composed exclusively of oxygen atoms. Allotropes must consist of only one element, so water cannot be considered an allotrope of oxygen.
C. Acidic oxide
“Acidic oxide” is not a specific substance but a general classification of oxides—usually nonmetal oxides—that react with water to form acids (such as sulphur dioxide or carbon dioxide). This term refers to a category of compounds, not a distinct molecular form of elemental oxygen, and therefore cannot be an allotrope.
D. Carbon dioxide
Carbon dioxide (CO₂) is a compound made of carbon and oxygen atoms. Because it contains more than one element, it does not meet the definition of an allotrope. Allotropes must be different structural forms of the same element only.
Conclusion
A triatomic allotrope of oxygen must consist of three oxygen atoms bonded together and contain no other elements. Ozone (O₃) meets this definition and
represents a distinct molecular form of elemental oxygen, different from the more common diatomic oxygen (O₂).
Topic Flashcards
Click to FlipWhat is the definition of an allotrope? What key feature distinguishes them from compounds?
Allotropes are different structural or molecular forms of the same element in the same physical state. The key feature is that they contain atoms of only one type of element.
What are the molecular formulas and common names for the two most familiar allotropes of oxygen?
Diatomic oxygen: O₂ (often just called "oxygen"). Triatomic oxygen: O₃ (called "ozone").
How do the chemical properties of ozone (O₃) differ from those of diatomic oxygen (O₂), and why is ozone a stronger oxidizing agent?
Ozone is much more reactive and a stronger oxidizing agent than O₂. Its O₃ structure is less stable, making it more eager to accept electrons from other substances to form the more stable O₂.
Name two other common elements that exhibit allotropy and give one example for each
1) Carbon: Graphite, Diamond, Graphene. 2) Phosphorus: White phosphorus, Red phosphorus.
Where is the ozone layer located in Earth's atmosphere, and what critical role does it play?
It is located in the stratosphere. It absorbs most of the sun's harmful ultraviolet (UV) radiation, protecting life on Earth.