What is the product of combustion of a hydrocarbon in excess oxygen?
-
A
Carbon dioxide and water
-
B
Naphthalene
-
C
Chlorine and bromine
-
D
Carbonium ions
The product of combustion of a hydrocarbon in excess oxygen is carbon dioxide and water.
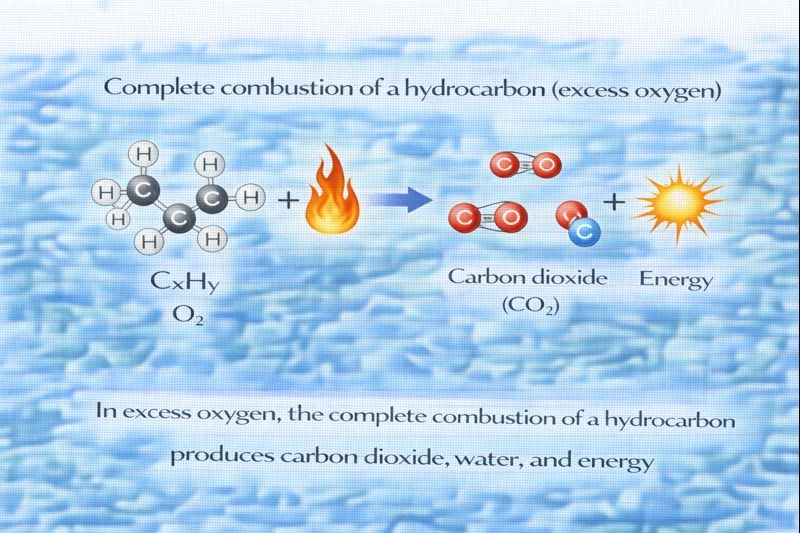
When a hydrocarbon reacts with an abundant supply of oxygen, the reaction is known as complete combustion. Hydrocarbons are compounds composed only of carbon and hydrogen atoms. During complete combustion, all carbon atoms are fully oxidized to carbon dioxide (CO₂), and all hydrogen atoms are oxidized to water (H₂O). This process releases a significant amount of energy, which is why hydrocarbons are commonly used as fuels.
A. Carbon dioxide and water
In excess oxygen, hydrocarbons undergo complete combustion, meaning there is sufficient oxygen to allow full oxidation of both carbon and hydrogen. Carbon atoms form carbon dioxide, and hydrogen atoms form water. The general equation for complete combustion is:
CₓHᵧ + O₂ → CO₂ + H₂O (balanced as required).
These products are stable, low-energy molecules, which is why combustion reactions release energy.
B. Naphthalene
Naphthalene is a polycyclic aromatic hydrocarbon composed of carbon and hydrogen atoms arranged in fused benzene rings. It is itself a hydrocarbon, not a product of combustion. Combustion breaks hydrocarbons down into simpler oxidized molecules rather than forming larger, more complex hydrocarbons like naphthalene.
C. Chlorine and bromine
Chlorine (Cl₂) and bromine (Br₂) are halogen elements and are not involved in the combustion of hydrocarbons unless specifically present as reactants. Combustion reactions involve hydrocarbons reacting with oxygen, not halogens, so these substances cannot be products of the reaction.
D. Carbonium ions
Carbonium ions (carbocations) are highly reactive, short-lived intermediates that appear in certain organic reaction mechanisms, such as rearrangements or substitution reactions. They are not stable end products and do not form during the complete combustion of hydrocarbons.
Conclusion
In the presence of excess oxygen, hydrocarbons undergo complete combustion, resulting in the full oxidation of carbon to carbon dioxide and hydrogen to water. These stable molecules are the characteristic products of complete combustion and explain why carbon dioxide and water are formed.
Topic Flashcards
Click to FlipWrite the general balanced chemical equation for the complete combustion of a hydrocarbon with the formula CₓHᵧ.
CₓHᵧ + (x + y/4)O₂ → xCO₂ + (y/2)H₂O (with appropriate coefficients).
What are the typical products of incomplete combustion of a hydrocarbon, and under what condition does it occur?
Carbon monoxide (CO) and/or carbon soot (C), along with water. It occurs when there is a limited supply of oxygen.
Why is complete combustion preferred in engines and heaters from both an energy efficiency and environmental standpoint?
Complete combustion releases more energy per mole of fuel and produces only CO₂ and H₂O, avoiding toxic CO and particulate pollution from soot.
How would you experimentally demonstrate that water is a product of hydrocarbon combustion?
By holding a cold, dry surface (like a beaker of ice water) above the flame; water vapor will condense into liquid droplets on the surface.
The combustion of 1 mole of propane (C₃H₈) releases 2220 kJ of energy. How much energy is released when 44 g of propane is completely burned?
2220 kJ. (44 g is the molar mass of propane, so 44 g = 1 mole, releasing 2220 kJ).