What is the name of the compound CH₃CH₂CH₂CH₃?
-
A
Cyclobutane
-
B
Butane
-
C
Butene
-
D
Butyne
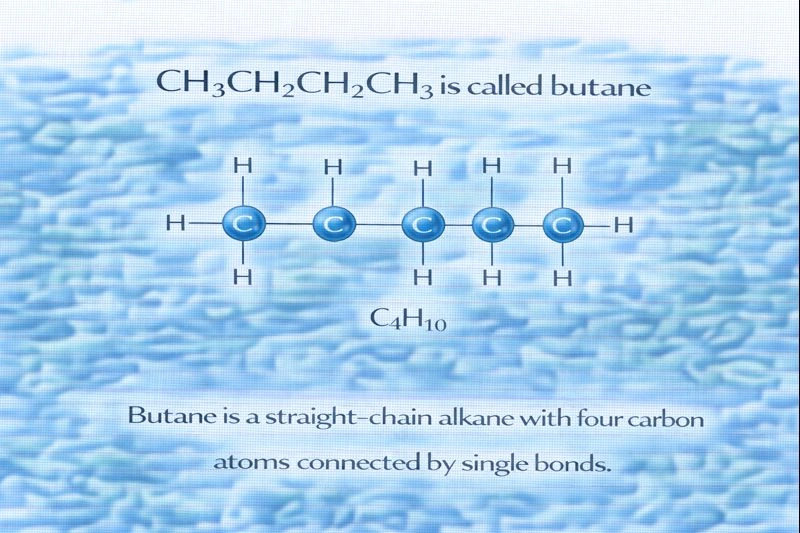
The compound CH₃CH₂CH₂CH₃ is called butane.
This chemical formula represents a straight-chain hydrocarbon composed of four carbon atoms connected by single bonds and surrounded by hydrogen atoms. When the carbon atoms are linked only by single covalent bonds and the molecule is fully saturated with hydrogen, the compound belongs to the alkane family. Alkanes follow the general molecular formula CₙH₂ₙ₊₂, and with four carbon atoms, the expected formula is C₄H₁₀, which matches CH₃CH₂CH₂CH₃.
A. Cyclobutane
Cyclobutane is a cyclic alkane, meaning its four carbon atoms are connected in a ring rather than in a straight chain. Because it forms a ring, cyclobutane has the molecular formula C₄H₈, not C₄H₁₀. The given structure is clearly linear, not cyclic, so it cannot be cyclobutane.
B. Butane
Butane is the straight-chain alkane with four carbon atoms. Its structure can be written as CH₃CH₂CH₂CH₃, and it follows the alkane naming rules: the prefix “but-” indicates four carbon atoms, and the suffix “-ane” indicates that all carbon–carbon bonds are single bonds. This makes butane a saturated hydrocarbon.
C. Butene
Butene is an alkene, which means it contains at least one carbon–carbon double bond. Alkenes follow the general formula CₙH₂ₙ, so butene has the formula C₄H₈. Since CH₃CH₂CH₂CH₃ contains only single bonds and has ten hydrogen atoms, it cannot be an alkene.
D. Butyne
Butyne is an alkyne, which contains a carbon–carbon triple bond. Alkynes follow the general formula CₙH₂ₙ₋₂, so butyne has the formula C₄H₆. The given compound does not contain a triple bond and has far more hydrogen atoms than an alkyne would.
Conclusion
The formula CH₃CH₂CH₂CH₃ represents a straight-chain, saturated hydrocarbon with four carbon atoms and only single bonds. Based on alkane naming rules and molecular structure, this compound is correctly identified as butane.
Topic Flashcards
Click to FlipWhat is the general molecular formula for an alkane, and how does butane (C₄H₁₀) fit this pattern?
The general formula is CₙH₂ₙ₊₂. For butane, n=4, so 2n+2 = (2*4)+2 = 10 hydrogens, matching C₄H₁₀.
What does the suffix "-ane" in the name "butane" indicate about the bonds between carbon atoms?
The suffix "-ane" indicates that all carbon-carbon bonds in the molecule are single bonds (it is a saturated hydrocarbon).
What is the structural difference between butane (CH₃CH₂CH₂CH₃) and isobutane ((CH₃)₃CH)?
Butane is a straight-chain (normal) alkane. Isobutane is a branched-chain isomer; it has three carbon atoms in a central carbon, forming a "T" shape.
Why can't the formula CH₃CH₂CH₂CH₃ belong to an alkene like butene or an alkyne like butyne?
Alkenes (C₄H₈) have a double bond and fewer hydrogens. Alkynes (C₄H₆) have a triple bond and even fewer hydrogens. The given formula has the maximum hydrogens for 4 carbons, indicating only single bonds.
If you saw the condensed formula (CH₃)₂CHCH₃, what is the name of this C₄H₁₀ isomer, and what type of chain does it have?
This is isobutane (or 2-methylpropane). It has a branched chain with a methyl (CH₃) group attached to the second carbon of a three-carbon propane chain.