Which of the following describes a typical gas?
-
A
Indefinite shape and indefinite volume
-
B
Indefinite shape and definite volume
-
C
Definite shape and definite volume
-
D
Definite shape and indefinite volume
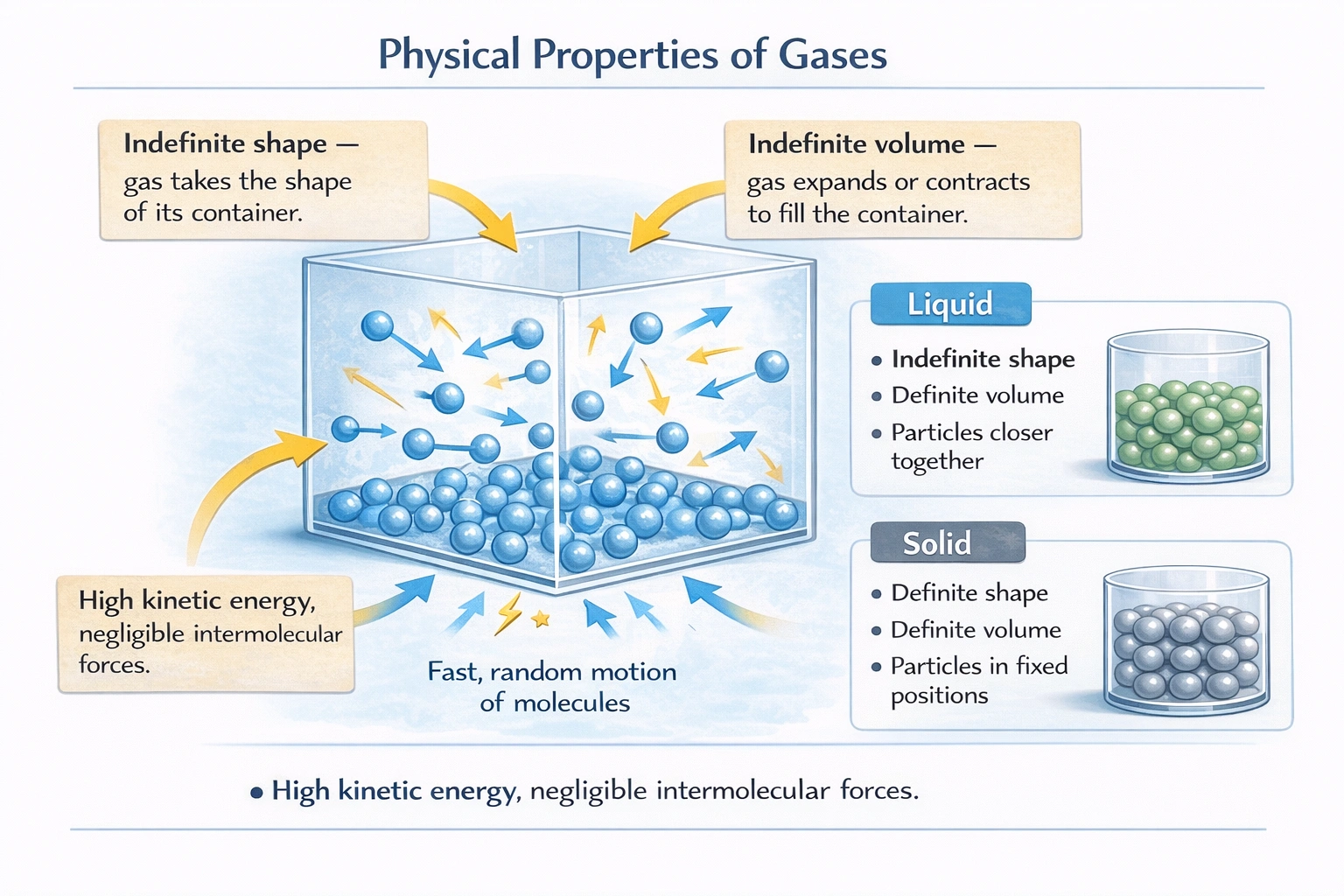
Gases possess indefinite shape and indefinite volume.
Gas molecules exhibit negligible intermolecular forces and high kinetic energy relative to thermal energy, enabling continuous random motion that distributes particles homogeneously throughout available volume producing no fixed boundaries, meniscus, or resistance to container shape changes.
A) Indefinite shape and indefinite volume
Gas molecules move freely with kinetic energies far exceeding intermolecular attraction energies, causing them to disperse until uniformly distributed throughout their container. A gas adopts the container's shape completely (indefinite shape) and expands or contracts to occupy the entire container volume (indefinite volume)—demonstrated when transferring gas between containers of different sizes: the gas fully fills each container without maintaining its previous volume. This behavior stems from the ideal gas law (PV = nRT), where volume directly depends on container dimensions at constant temperature and pressure.
B) Indefinite shape and definite volume
This describes liquids, which conform to container shape below the meniscus (indefinite shape) but maintain constant volume regardless of container size due to stronger intermolecular forces limiting molecular separation. Pouring 100 mL of water into containers of varying widths produces different shapes but identical volumes—behavior impossible for gases, which would expand to fill the entire container volume.
C) Definite shape and definite volume
This characterizes crystalline solids, where strong intermolecular or ionic bonds lock particles into fixed positions within a rigid lattice. Solids maintain both shape and volume independent of container geometry—evident when placing a sugar cube in a beaker: it retains cubic form and volume without conforming to the beaker's curvature. Amorphous solids (e.g., glass) also maintain definite volume though lacking long-range order.
D) Definite shape and indefinite volume
No common state of matter exhibits this combination. Definite shape requires structural rigidity from strong intermolecular forces, which simultaneously restrict volume changes—making indefinite volume physically incompatible with shape maintenance. Hypothetical materials with this property would violate thermodynamic principles governing condensed phases.
Conclusion:
The kinetic molecular theory explains gaseous behavior through high particle velocities, negligible intermolecular forces, and elastic collisions that distribute molecules uniformly throughout available space. This produces the dual indefiniteness of shape and volume—fundamental properties distinguishing gases from liquids (indefinite shape, definite volume) and solids (definite shape and volume). Option A correctly identifies these defining characteristics, essential for understanding gas laws, atmospheric behavior, and respiratory physiology where air conforms completely to pulmonary airway geometry and alveolar volume.