Which substance’s volume would be most affected by temperature change?
-
A
Liquid nitrogen
-
B
Salt crystals
-
C
Hydrogen gas
-
D
Iron filings
The substance whose volume would be most affected by temperature change is hydrogen gas.
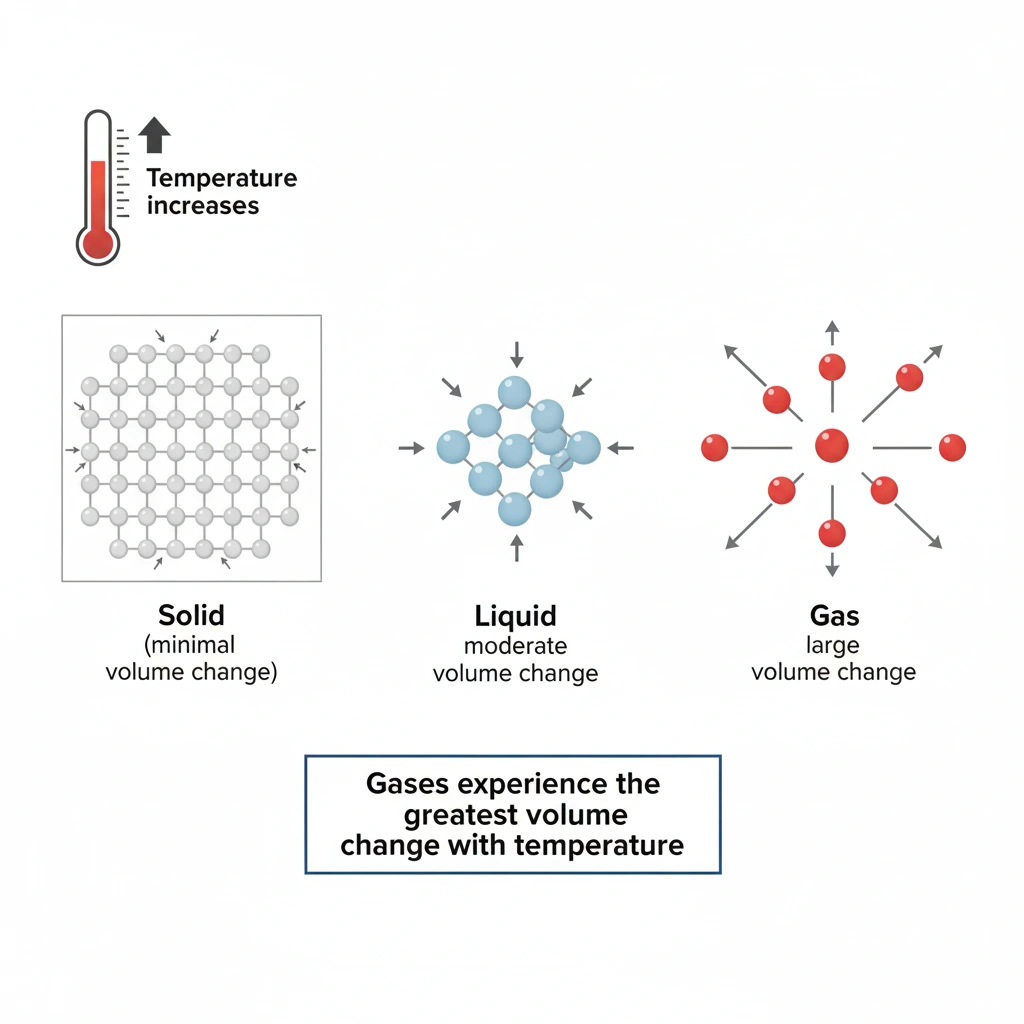
The effect of temperature on volume depends strongly on the state of matter. Gases are far more sensitive to temperature changes than liquids or solids because the particles in a gas are much farther apart and move more freely. As temperature increases, gas particles move faster and spread out, causing a significant change in volume.
A) Liquid nitrogen
Liquids do expand when heated, but their particles are still relatively close together. As a result, the change in volume with temperature is moderate and much smaller than that observed in gases.
B) Salt crystals
Salt is a solid, and solids exhibit very little thermal expansion. The rigid lattice structure of a solid restricts particle motion, so changes in temperature produce only minimal changes in volume.
C) Hydrogen gas
Gases follow Charles’s law, which states that volume is directly proportional to absolute temperature at constant pressure. A small increase in temperature leads to a relatively large increase in volume. Hydrogen gas, like all gases, has a much greater volumetric expansion per degree of temperature change than liquids or solids.
D) Iron filings
Iron is a solid metal and expands only slightly with temperature. Even with noticeable heating, the change in volume of iron is negligible compared with that of a gas.
Conclusion
Among solids, liquids, and gases, gases experience the greatest volume change for a given temperature change. Therefore, hydrogen gas is the substance whose volume is most affected by temperature.
Topic Flashcards
Click to FlipState Charles's Law. What are the conditions (which variables must be held constant)?
Charles's Law states that the volume (V) of a fixed amount of gas is directly proportional to its absolute temperature (T). In formula: V ∝ T (when pressure and amount of gas are constant).
Rank solids, liquids, and gases in order from least to greatest thermal volume expansion for a given temperature increase. Explain why.
Solids < Liquids < Gases. Solids have strong intermolecular forces in a rigid structure, liquids have moderate forces, and gases have very weak forces with particles far apart, allowing much greater expansion.
If a sample of hydrogen gas at 300 K occupies 2.0 L, what volume will it occupy at 450 K (assuming constant pressure)?
Using Charles's Law (V₁/T₁ = V₂/T₂): V₂ = V₁ * (T₂/T₁) = 2.0 L * (450 K / 300 K) = 2.0 L * 1.5 = 3.0 L.
Why does a gas show a much larger volume change with temperature compared to a solid? Use ideas from the kinetic molecular theory.
Gas particles are far apart with negligible attractive forces. Increasing temperature greatly increases their average kinetic energy, causing more frequent, forceful collisions with the container walls, which pushes the walls outward, increasing volume significantly. In solids, particles are locked in place and only vibrate more.
What is the coefficient of volume expansion, and how does its typical magnitude compare for gases versus solids?
It's a constant (β) that quantifies the fractional change in volume per degree of temperature change (ΔV/V₀ = βΔT). For ideal gases, β is about 0.00367 K⁻¹ (or 1/273 K⁻¹), while for solids like iron, it's on the order of 0.000036 K⁻¹—about 100 times smaller.