What is a cation?
-
A
A positively charged ion
-
B
A negatively charged ion
-
C
A positively charged covalent bond
-
D
A negatively charged covalent bond
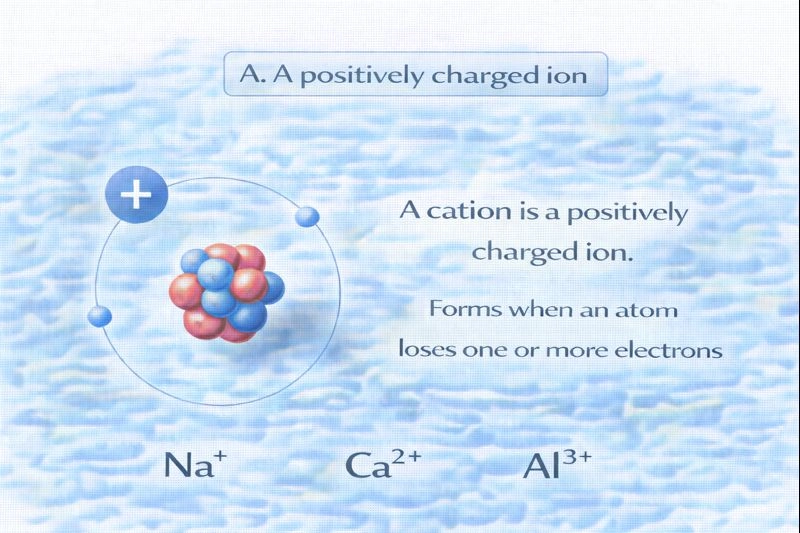
A cation is a positively charged ion.
Ions are formed when atoms gain or lose electrons. Because electrons carry a negative charge, losing electrons causes an atom to become positively charged. This type of ion is called a cation.
A. A positively charged ion
A cation forms when a neutral atom loses one or more electrons. The loss of negatively charged electrons leaves the atom with more protons than electrons, giving it a net positive charge. Common examples include sodium ions (Na⁺), calcium ions (Ca²⁺), and aluminum ions (Al³⁺).
B. A negatively charged ion
A negatively charged ion is called an anion, not a cation. Anions form when atoms gain electrons, such as chloride (Cl⁻) or oxide (O²⁻).
C. A positively charged covalent bond
Covalent bonds involve the sharing of electrons between atoms. While a molecule can carry an overall charge, individual covalent bonds themselves are not classified as positively or negatively charged ions.
D. A negatively charged covalent bond
This option is also incorrect. As with option C, covalent bonds are not ions and are not described in terms of overall positive or negative charge.
Conclusion
A cation is an ion that carries a positive charge due to the loss of one or more electrons.
Topic Flashcards
Click to FlipHow does a neutral atom become a cation? What happens to its electrons?
It loses one or more electrons. This leaves the atom with more protons than electrons, resulting in a net positive charge.
Which group on the periodic table (Group 1, 2, 16, or 17) is most likely to form cations, and what is a typical charge?
Groups 1 and 2 (alkali and alkaline earth metals). Group 1 typically forms 1+ cations (e.g., Na⁺), and Group 2 forms 2+ cations (e.g., Mg²⁺).
What is the difference between a sodium atom (Na) and a sodium cation (Na⁺) in terms of their electron configuration?
A neutral Na atom has the configuration 1s²2s²2p⁶3s¹ (11 electrons). The Na⁺ cation loses the single 3s electron, leaving it with the configuration 1s²2s²2p⁶ (10 electrons, isoelectronic with neon).
If an aluminum atom (Al, atomic number 13) loses three electrons to form a cation, what is its charge and symbol?
It forms an Al³⁺ cation. (13 protons, 10 electrons, charge = +3).
In an ionic compound like NaCl, which ion is the cation and what is its role in the compound's formation?
Na⁺ is the cation. It forms when sodium donates an electron to chlorine, leading to an electrostatic attraction (ionic bond) with the Cl⁻ anion.