What is the correct formula for sodium nitrate?
-
A
Na₂NO
-
B
Na₃NO
-
C
NaNO₂
-
D
NaNO₃
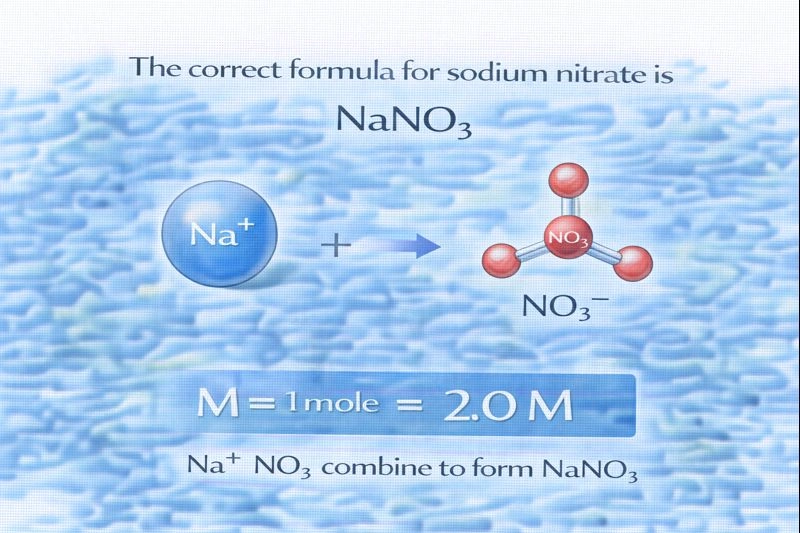
The correct formula for sodium nitrate is NaNO₃.
Sodium nitrate is an ionic compound formed from sodium ions (Na⁺) and nitrate ions (NO₃⁻). When writing the formula for an ionic compound, the total positive charge must balance the total negative charge so that the compound is electrically neutral. Because sodium carries a +1 charge and the nitrate ion carries a −1 charge, they combine in a one-to-one ratio to form sodium nitrate.
A. Na₂NO
This formula suggests the presence of two sodium ions and an anion written as NO, which would imply an unusual and non-existent polyatomic ion with a −2 charge. There is no stable or recognized polyatomic ion with the formula NO²⁻ in basic chemical nomenclature. Therefore, this formula does not represent sodium nitrate.
B. Na₃NO
This formula would require an anion with a −3 charge to balance the three sodium ions (each +1). No such common polyatomic ion written as NO³⁻ exists. As a result, this formula does not correspond to any known sodium compound and is not chemically valid for sodium nitrate.
C. NaNO₂
NaNO₂ is the chemical formula for sodium nitrite, not sodium nitrate. The nitrite ion has the formula NO₂⁻, while the nitrate ion has the formula NO₃⁻. Although the names nitrite and nitrate are similar, they represent different polyatomic ions with different numbers of oxygen atoms and different chemical properties.
D. NaNO₃
This option is correct. The nitrate ion (NO₃⁻) has a −1 charge, and the sodium ion (Na⁺) has a +1 charge. Combining one sodium ion with one nitrate ion results in a neutral compound with the formula NaNO₃, which is the correct and accepted formula for sodium nitrate.
Conclusion
Sodium nitrate is formed by combining Na⁺ ions with NO₃⁻ ions in a one-to-one ratio to achieve electrical neutrality. It is important to distinguish nitrate (NO₃⁻) from nitrite (NO₂⁻), as they form different compounds. Sodium nitrate is correctly written as NaNO₃.
Topic Flashcards
Click to FlipWhat are the charges on the sodium ion and the nitrate ion, and why does this lead to the formula NaNO₃?
Sodium ion is Na⁺ (+1 charge). Nitrate ion is NO₃⁻ (-1 charge). To balance the charges (+1 and -1), one of each ion combines, giving the formula NaNO₃.
What is the systematic process for writing the formula of an ionic compound like sodium nitrate?
1. Identify the ions (Na⁺ and NO₃⁻). 2. Determine their charges. 3. Use the smallest whole number ratio of ions that makes the total positive and negative charge equal (1:1 for NaNO₃).
What is the difference between a nitrate ion and a nitrite ion, both in formula and name of the sodium compound each forms?
Nitrate is NO₃⁻ (forms sodium nitrate, NaNO₃). Nitrite is NO₂⁻ (forms sodium nitrite, NaNO₂). They differ by one oxygen atom.
Using the criss-cross method, show how you would derive the formula for sodium nitrate from the ions Na⁺ and NO₃⁻.
The charges are +1 and -1. The subscript for Na becomes the absolute value of the nitrate's charge (1). The subscript for NO₃ becomes the absolute value of sodium's charge (1). This gives Na₁(NO₃)₁, simplified to NaNO₃.
If you combined sodium ions with phosphate ions (PO₄³⁻), what would be the formula of the resulting ionic compound?
Na₃PO₄. (Three Na⁺ ions, each +1, are needed to balance the -3 charge of one phosphate ion).