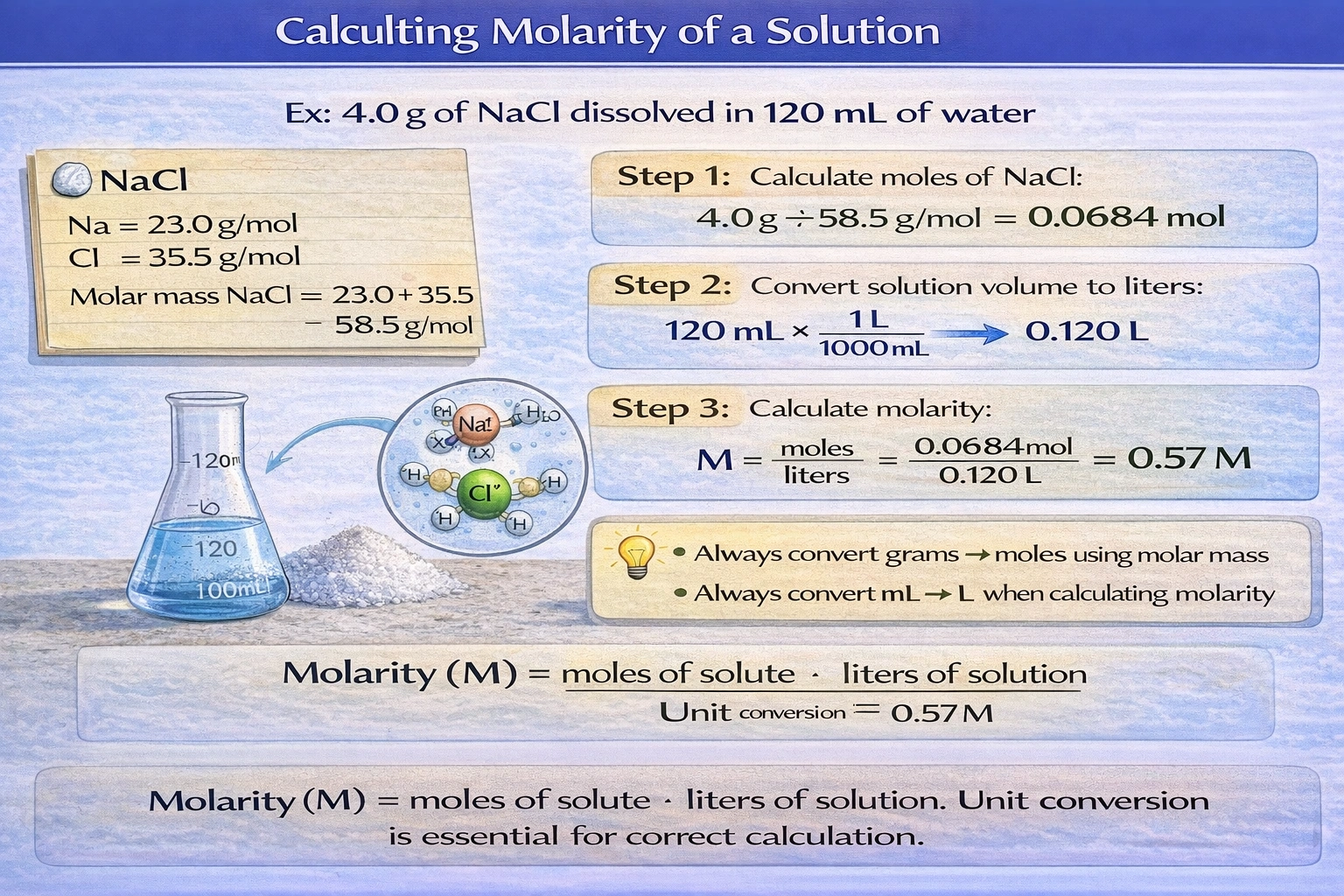
What is the molarity of a solution made by dissolving 4.0 grams of NaCl into enough water to make 120 mL of solution? The atomic mass of Na is 23.0 g/mol and Cl is 35.5 g/mol.
-
A
0.34 M
-
B
0.57 M
-
C
0.034 M
-
D
0.057 M
The molarity equals 0.57 M, calculated by determining moles of NaCl (4.0 g ÷ 58.5 g/mol = 0.0684 mol) and dividing by solution volume in liters (0.120 L), yielding 0.57 mol/L.
Molarity (M) represents moles of solute per liter of solution, requiring conversion of mass to moles using molar mass and volume to liters before division—fundamental stoichiometric calculations essential for solution preparation in chemistry and biology.
A) 0.34 M
This value might result from incorrectly using volume as 0.240 L (doubling 120 mL) or miscalculating molar mass as 117 g/mol (doubling correct value). Verification: 0.0684 mol ÷ 0.240 L = 0.285 M (not 0.34); 4.0 g ÷ 117 g/mol = 0.0342 mol ÷ 0.120 L = 0.285 M. Neither path yields 0.34 M, suggesting possible arithmetic error in intermediate steps.
B) 0.57 M
Molar mass of NaCl = 23.0 + 35.5 = 58.5 g/mol. Moles of NaCl = mass ÷ molar mass = 4.0 g ÷ 58.5 g/mol = 0.068376 mol. Volume in liters = 120 mL × (1 L/1000 mL) = 0.120 L. Molarity = moles ÷ liters = 0.068376 mol ÷ 0.120 L = 0.5698 M, which rounds to 0.57 M with two significant figures (matching the precision of 4.0 g and 120 mL inputs). This calculation correctly applies molarity definition with proper unit conversions.
C) 0.034 M
This value equals approximately half of option A and might result from dividing moles by volume in milliliters without conversion: 0.0684 mol ÷ 120 mL = 0.00057 mol/mL, which equals 0.57 mol/L—not 0.034. Alternatively, it could stem from using 4.0 g ÷ 58.5 g/mol = 0.068 mol then incorrectly dividing by 2 (perhaps confusing with dilution factor). The value 0.034 appears as half of 0.068, suggesting arithmetic halving error.
D) 0.057 M
This equals exactly one-tenth of the correct answer, likely resulting from volume conversion error: using 1200 mL instead of 0.120 L (0.0684 mol ÷ 1.20 L = 0.057 M) or misplacing decimal in volume conversion (120 mL = 1.20 L instead of 0.120 L). Such decimal errors commonly occur when converting between milliliters and liters without careful attention to the 1000:1 ratio.
Conclusion:
Molarity calculations require meticulous attention to unit conversions and significant figures. The correct solution involves three sequential steps: (1) sum atomic masses for molar mass (58.5 g/mol), (2) convert mass to moles (0.0684 mol), and (3) convert volume to liters before division (0.57 M). Option B correctly executes all steps with appropriate rounding. Options A, C, and D reflect common student errors: volume miscalculation (A), decimal placement errors (D), or arithmetic mistakes (C). Mastery of these foundational calculations proves essential for laboratory work in chemistry, biology, and medicine—where solution concentrations directly impact reaction rates, osmotic balance, and pharmacological efficacy.