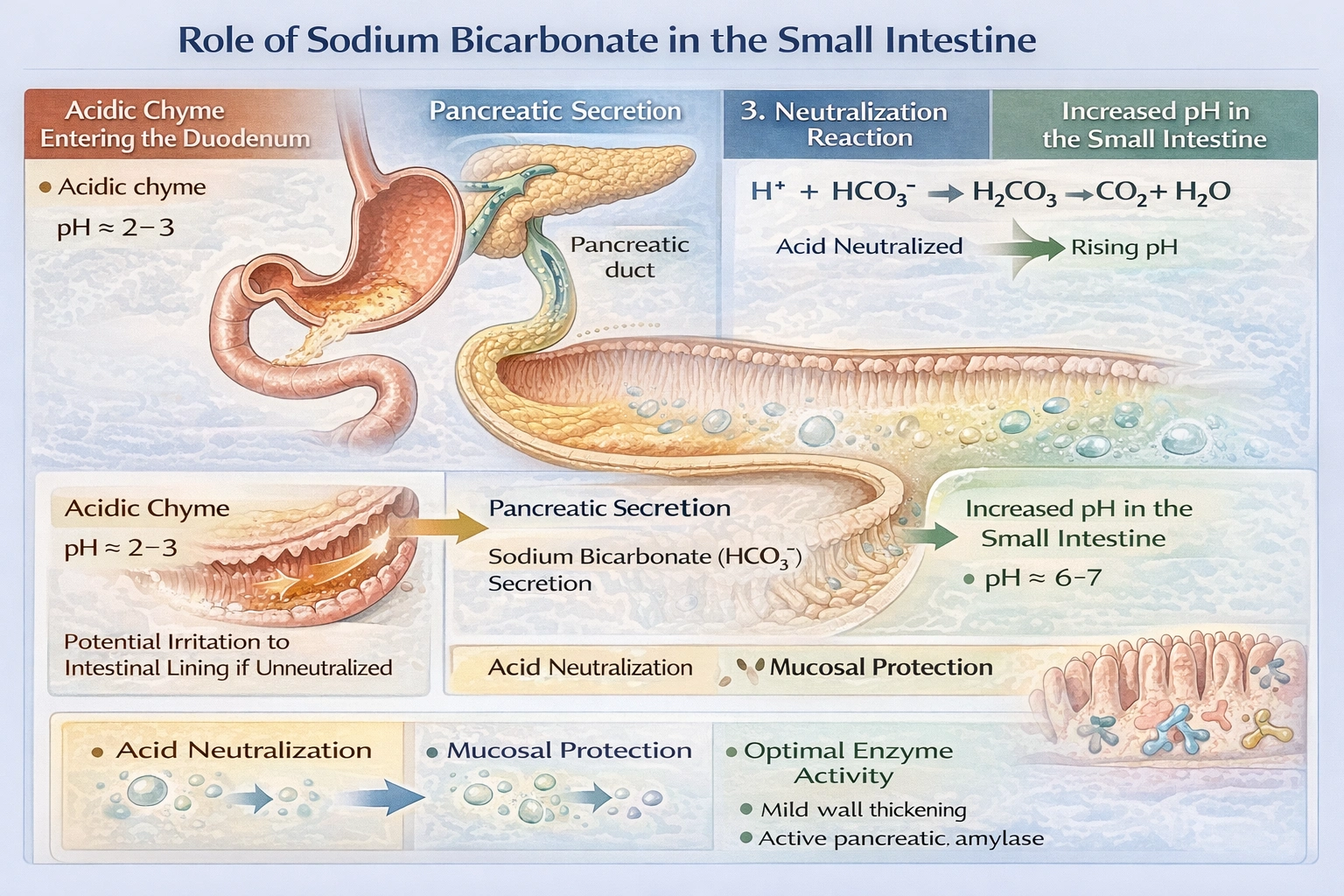
What is the purpose of sodium bicarbonate when released into the lumen of the small intestine?
-
A
It works to chemically digest fats in the chyme.
-
B
It decreases the pH of the chyme so as to prevent harm to the intestine.
-
C
It works to chemically digest proteins in the chyme.
-
D
It increases the pH of the chyme so as to prevent harm to the intestine
Sodium bicarbonate increases the pH of the chyme so as to prevent harm to the intestine.
Pancreatic duct cells secrete bicarbonate-rich fluid that neutralizes acidic gastric chyme entering the duodenum, raising luminal pH from approximately 2–3 to 6–7 to protect mucosal integrity and enable optimal pancreatic enzyme function.
A) It works to chemically digest fats in the chyme
Fat digestion occurs through pancreatic lipase and bile salt emulsification, not bicarbonate. Bicarbonate creates the neutral pH environment required for lipase activity but does not itself hydrolyze triglycerides.
B) It decreases the pH of the chyme so as to prevent harm to the intestine
Bicarbonate functions as a base that accepts hydrogen ions (H⁺ + HCO₃⁻ → H₂CO₃ → CO₂ + H₂O), thereby increasing pH through neutralization. Decreasing pH would make chyme more acidic and exacerbate mucosal damage risk.
C) It works to chemically digest proteins in the chyme
Protein digestion results from proteolytic enzymes including trypsin, chymotrypsin, carboxypeptidase, and aminopeptidases. Bicarbonate elevates pH to the neutral range required for these enzymes' optimal activity but lacks proteolytic capability itself.
D) It increases the pH of the chyme so as to prevent harm to the intestine
Gastric chyme enters the duodenum highly acidic due to hydrochloric acid secretion. Without neutralization, this acidity would erode the duodenal mucosa and inactivate pancreatic enzymes that function optimally near pH 7. Bicarbonate secretion from pancreatic duct cells and Brunner's glands raises luminal pH, protecting epithelial cells and enabling efficient enzymatic digestion.
Conclusion:
Sodium bicarbonate serves a critical protective and permissive role in intestinal physiology by neutralizing gastric acid. This pH elevation prevents mucosal injury and establishes the chemical environment necessary for pancreatic enzymes to complete macronutrient digestion.