What is the normal pH of blood?
-
A
7.25-7.35
-
B
7.35-7.45
-
C
7.0-7.5
-
D
7.50-7.65
The normal pH of human blood is 7.35–7.45.
Blood is slightly alkaline, and this narrow pH range is essential for normal cellular metabolism, enzyme activity, oxygen transport, and overall physiological stability. The body tightly regulates blood pH using buffer systems (especially the bicarbonate–carbonic acid system), the lungs (by controlling carbon dioxide levels), and the kidneys (by excreting or conserving hydrogen and bicarbonate ions).
A) 7.25–7.35
This range is lower than normal and indicates increased acidity in the blood.
When blood pH falls below 7.35, the condition is called acidemia. This can occur due to respiratory causes (such as hypoventilation leading to carbon dioxide retention) or metabolic causes (such as diabetic ketoacidosis or kidney failure).
Although values in this range may be seen in illness, they do not represent normal physiological conditions. Therefore, this option is incorrect.
B) 7.35–7.45
This is the normal physiological pH range of human blood.
Within this range, enzymes function optimally, oxygen binds efficiently to hemoglobin, and cellular chemical reactions proceed normally. Even small deviations outside this interval can impair organ function and disrupt metabolic processes.
Because this range accurately represents healthy arterial blood pH, this option is correct.
C) 7.0–7.5
This range is too broad and includes values that are dangerously acidic.
A pH near 7.0 indicates severe acidemia, which can cause cardiac dysfunction, decreased responsiveness of enzymes, and impaired oxygen delivery to tissues. Such levels are associated with life-threatening conditions rather than normal physiology.
Therefore, this option does not correctly describe normal blood pH.
D) 7.50–7.65
This range is above normal and represents alkalemia.
Blood pH values higher than 7.45 indicate alkalosis, which may result from hyperventilation, excessive loss of stomach acid, or certain metabolic disorders. Alkalosis can cause neuromuscular irritability, muscle cramps, cardiac arrhythmias, and reduced cerebral blood flow.
Because this range is not normal and reflects pathological conditions, it is incorrect.
Conclusion:
Normal blood pH is maintained within a very narrow range to support vital biochemical and physiological processes. Values below 7.35 indicate acidemia, and values above 7.45 indicate alkalemia.
Therefore, the correct normal blood pH range is 7.35–7.45.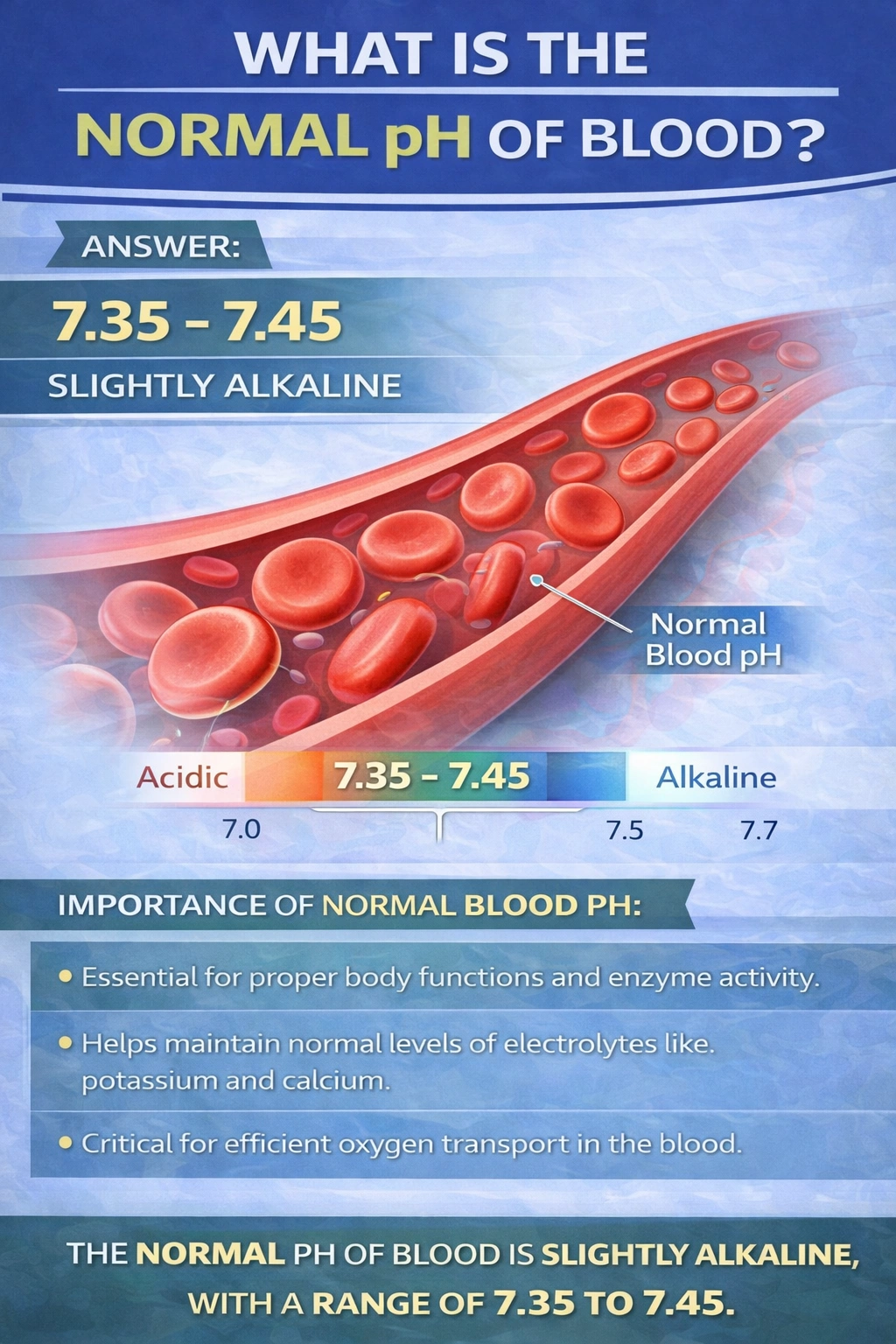
Topic Flashcards
Click to FlipWhat is the primary buffer system in the blood that maintains pH within the normal range?
The bicarbonate–carbonic acid buffer system ( HCO 3 − / H 2 CO 3 HCO 3 − /H 2 CO 3 ).
Which organ primarily regulates blood pH in the short term by controlling the exhalation of CO₂?
The lungs (through respiratory rate and depth).
What is the medical term for a blood pH below 7.35?
Acidemia (or acidosis, referring to the process causing it).
Which organ plays the major long-term role in pH regulation by excreting or reabsorbing hydrogen ions ( H + H + ) and bicarbonate ( HCO 3 − HCO 3 − )?
The kidneys.
What is the condition called when blood pH rises above 7.45, often due to hyperventilation?
Alkalemia (or respiratory alkalosis if caused by low CO₂).